Chalcogen Bonding in Catalysis
Weak noncovalent interactions are the key to tailor make complex chemical systems to realize a specific function. Our most recent efforts geared towards expanding the toolbox of noncovalent interactions resulted in the application of chalcogen bonds to transmembrane transport and catalysis.1,2 Chalcogen bonds arise when the electron deficient σ* orbitals, on sulfur, selenium or tellurium interact with a lewis-basic partner. Ideal spatial orientation of the sigma holes led us to select the scaffold of Dithieno[3,2-b;2’,3’-d]thiophenes (DTTs) for bidentate chalcogen binding
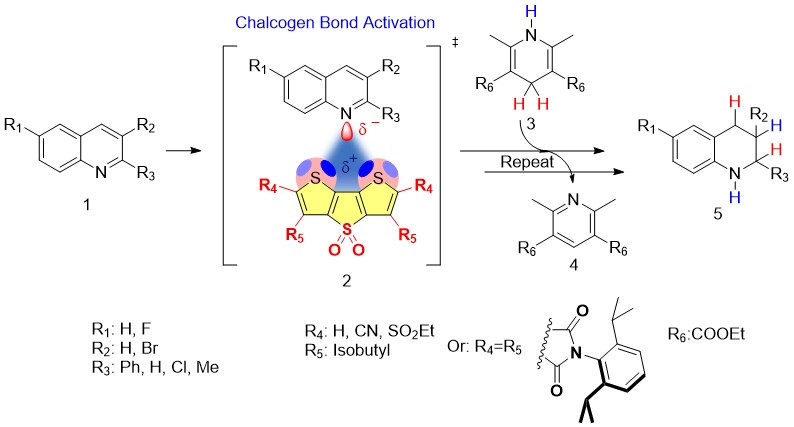
Through interaction with the nitrogen lone pair DTTs are a privileged motive to activate imines and quinolines for Hantzsch ester mediated transfer hydrogenation. Over a 1000-fold rate enhancement, stronger activities with deeper σ holes and wider bite angles, chloride inhibition and correlation with computed binding strengths yield strong evidence for operational chalcogen bonds. Lessons learned are being currently applied to asymmetric catalysis with selenium based chalcogen binders.
[1] Sebastian Benz, Mariano Macchione, Quentin Verolet, Jiri Mareda, Naomi Sakai, Stefan Matile, J. Am. Chem. Soc., 2016, 138, 9093–9096.
[2] Sebastian Benz, Javier López-Andarias, Jiri Mareda, Naomi Sakai, Stefan Matile, Angew. Chem. Int. Ed., 2017, 56, 812–815.