New entries into amino-benzonorbornene chemistry
Cationic rearrangements based routes, from early Process Research towards two key intermediates of fungicidal active ingredients, will be disclosed. The synthesis of functionalized benzonorbonenes from cycloaddition between cyclopentadiene and benzynes, followed by rearrangements to adjust the substitution pattern and the functional groups, will be presented from the point of view of route scouting and novel reactions assessments.
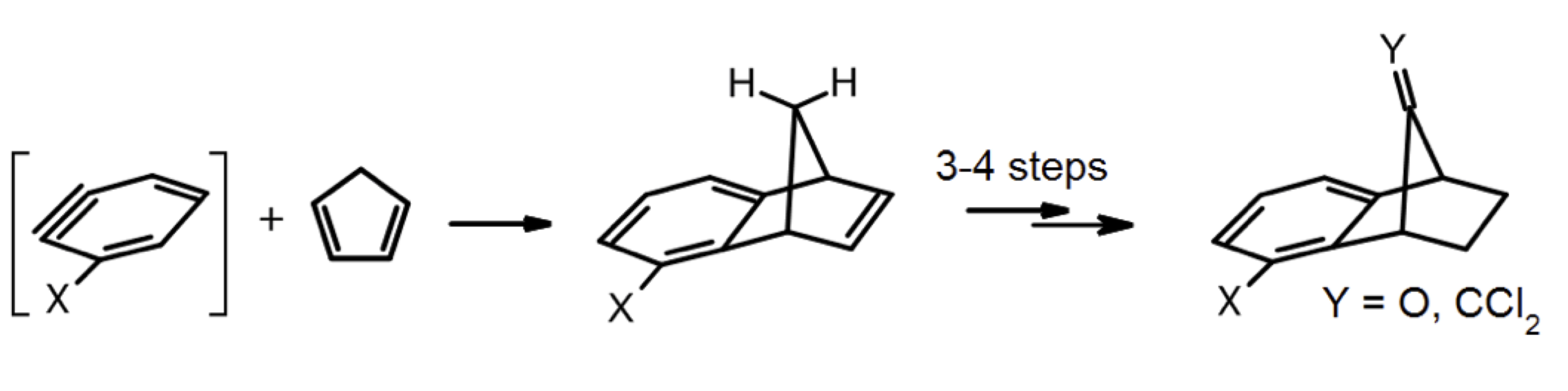
The chemistry developed for targeting specific amino-benzonorbornene derivatives[1,2] will be broadened to, and exemplified by, other substrates. Rare or unprecedented electrophilic acylations and alkylations of double bonds by cations, triggering in situ further cationic 1,2-Wagner-Meerwein shifts will be disclosed; in both cyclic and acyclic series, [1,2]-shifts of aromatic rings led by β-cation stabilisation by silicon groups will also be exemplified.
[1] R. Dumeunier, H. Tobler, PCT Int. Appl. (2011), WO/2011/131543.
[2] R. Dumeunier, F. Schleth, T. Vettiger, M. Rommel, S. Trah, PCT Int. Appl. (2011), WO/2011/131545.