Protein-nucleotide interactions studied by magnetic resonance
In this contribution we present strategies to characterize protein-nucleotide interactions in large biological assemblies using the example of the (double-) hexameric DnaB helicase from Helicobacter pylori with a molecular weight of 672 kDa. The binding of nucleotides (ATP-analogues and single-stranded DNA, ssDNA) to the helicase is monitored by 31P,1H cross-polarization experiments. The observed 31P chemical-shift values allow a clear discrimination between the different nucleotides used in this work and reveal that two nucleotides of bound ssDNA can be structurally distinguished. 15N,13C correlation experiments highlight the role of arginine sidechains in binding to single-stranded DNA.
We further present dipolar-coupling based polarization transfer experiments from 31P spins of bound nucleotides to 13C or 15N spins of the protein (or vice versa) to describe nucleotide binding on a molecular level (see Figure 1). Residues involved in protein-nucleotide interactions are identified in CHHP and NHHP correlation experiments.
While conventional NMR experiments on such large protein assemblies typically suffer from low signal-to-noise ratios and thus long experimental measurements times, we used Dynamic Nuclear Polarization (DNP) enhanced MAS experiments to increase the sensitivity. The samples for DNP were prepared in the absence of glycerol which results in the highest sensitivity for DnaB. The obtained spectra allow to identify spin systems of the helicase in the neighborhood of 31P spins of the nucleotides. The extensive line-broadening at cryogenic temperatures is not a severe issue in this case due to the sparsity of the obtained spectra.
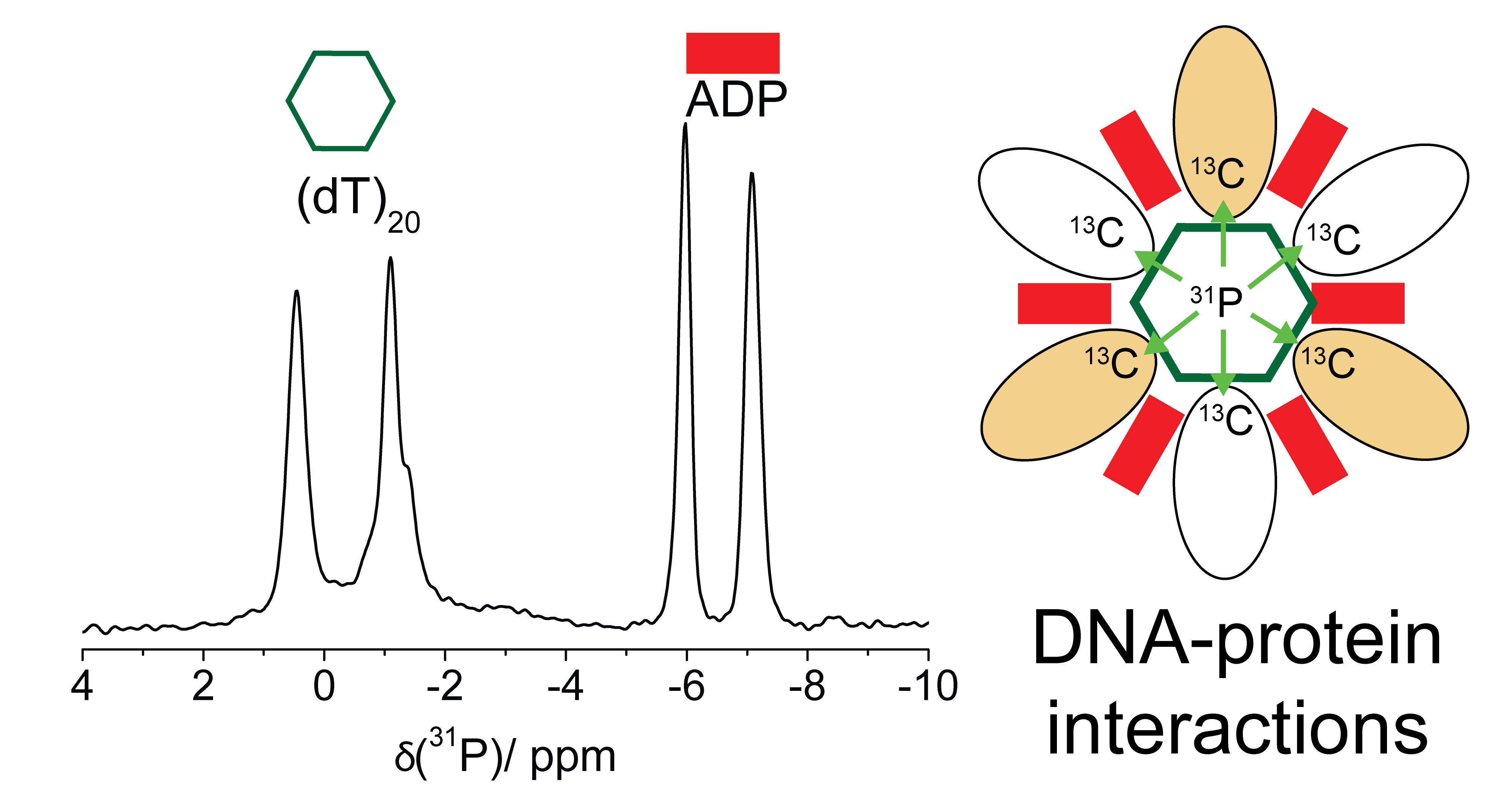
Figure 1: 31P spins of bound nucleotides can be used in solid-state NMR experiments to identify helicase-nucleotide interactions.