Palladium-Catalyzed Long-Range Deconjugative Isomerization of Highly Substituted α,β-Unsaturated Carbonyl Compounds
The long range isomerization/refunctionalization of olefins has emerged as an effective method for the construction of functionalized molecules. This redox neutral methodology relies on the use of transition metal complexes, with the economic and environmental advantage to avoid the formation of stoichiometric waste.[1] The main challenges for the successful development of such processes are (i) the difficult coordination of highly substituted (prochiral) olefins with metal catalysts,[2] severely narrowing the scope of these methodologies, and (ii) the control of the regioselectivity of metal hydride insertion across the C=C bond.[3]
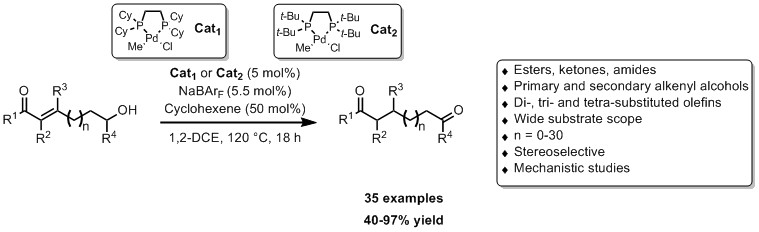
Building on previous studies in our group,[4] we report herein the application of two Pd catalysts to the deconjugation, isomerization and refunctionalization of α,β-unsaturated carbonyls in good to high yields.[5] Our system isomerizes di-, tri- and tetra-substituted olefins to highly valuable aldehydes and ketones regardless the chain length (>35 examples). Preliminary mechanistic will also be presented.
[1] a) Alexandre Vasseur, Jeffrey Bruffaerts, Ilan Marek, Nat. Chem. 2016, 8, 209-219; b) Timo Witt, Florian Stempfle, Philipp Roesle, Manuel Häußler, Stefan Mecking, ACS Catal. 2015, 5, 4519-4529; c) Tian-Sheng Mei, Harshkumar H. Patel, Matthew S. Sigman, Nature 2014, 508, 340-344; d) Hideaki Wakamatsu, Mayumi Nishida, Naoko Adachi, Miwako Mori, J. Org. Chem. 2000, 65, 3966-3970
[2] Taro Hamasaki, Fumitoshi Kakiuchi, Takuya Kochi, Chem. Lett. 2016, 45, 297-299
[3] Chun Zhang, Celine B. Santiago, Lei Kou, Matthew S. Sigman, J. Am. Chem. Soc. 2015, 137, 7290-7293
[4] Evgeny Larionov, Luqing Lin, Laure Guénée, Clément Mazet, J. Am. Chem. Soc. 2014, 136, 16882-16894
[5] Luqing Lin, Ciro Romano, Clément Mazet, J. Am. Chem. Soc. 2016, 138, 10344-10350