Rational design of sulfur-tolerant ruthenium catalysts for dry biomass derived CO methanation
The process of methane production from dry biomass, called wood-to-Synthetic Natural Gas (SNG), consists of 4 main steps: biomass gasification, syngas cleaning to remove catalyst poisons such as H2S, COS and C4H4S using “cold” gas cleaning technologies, methanation and upgrading to remove H2O and CO2.1 To make SNG cost-competitive, the concept of integrating gas cleaning with methanation, which utilizes the ability of ruthenium-based catalysts to be regenerated under oxidizing atmosphere after sulfur poisoning is explored.2
However, a complete recovery of the catalytic activity after the regeneration cannot be achieved so far, probably because of a combination of several reasons. Firstly, Al2O3 support of the nanoparticles can “store” some of the sulfur poisons in the form of sulfate, which prevent efficient regeneration.2 Secondly, TEM and XAS analysis evidences particle sintering upon recycling of originally 1 nm particle in Ru/Al2O3.
Here, we show that silica largely improve the regeneration process, because it is less prompt to sulfur storage (Fig. 1a). Operando XAS at the sulfur K-edge (Fig. 1b) and DRIFTS (Fig. 1c) showed that sulfate species formed on SiO2 are unstable and could be removed by subsequent treatment with H2. However, sintering still remains an issue. In addition, DRIFT spectroscopy revealed altered CO adsorption profile for the regenerated catalyst, implying that structural and/or electronic properties of the catalyst are changed after a poisoning-regeneration cycle.
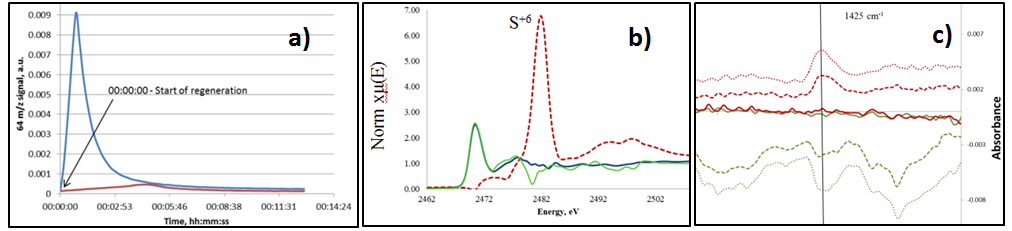
Figure 1: a) SO2 detected at the reactor outlet for Ru/Al2O3 (red) and Ru/SiO2 (blue) b) Sulfur K-edge XAS spectrum of Ru/SiO2 catalyst taken during methanation with poisoning (blue), regeneration in 1% O2 (red) and subsequent methanation (green); c) DRIFTS spectrum of Ru/SiO2 showing sulfate (ca. 1425 cm-1 band)4 formation (1% O2 regeneration after catalyst poisoning, red) and decomposition (1% H2, green) onSiO2 surface
[1] C. F. J. König et al. in T. J. Schildhauer, S. M. A. Biollaz (Eds.), Synthetic Natural Gas from Coal, Dry Biomass, and Power-to-Gas Applications, John Wiley & Sons, 2016.
[2] C. F. J. König et al., Catalysis Today, 2014, 229, 56 – 63
[3] D. L. Trimm in G. Ertl, H. Knözinger, J. Weitkamp (Eds.), Handbook of Heterogeneous Catalysis, VCH Verlagsgesellschaft mbH, 1997
[4] D. Bounechada, et al, J. Phys. Chem. C , 118, 2014 pp. 29713−29723