High-Pressure Synthesis and Characterization of β-GeSe — A Six-Membered-Ring Semiconductor in an Uncommon Boat Conformation
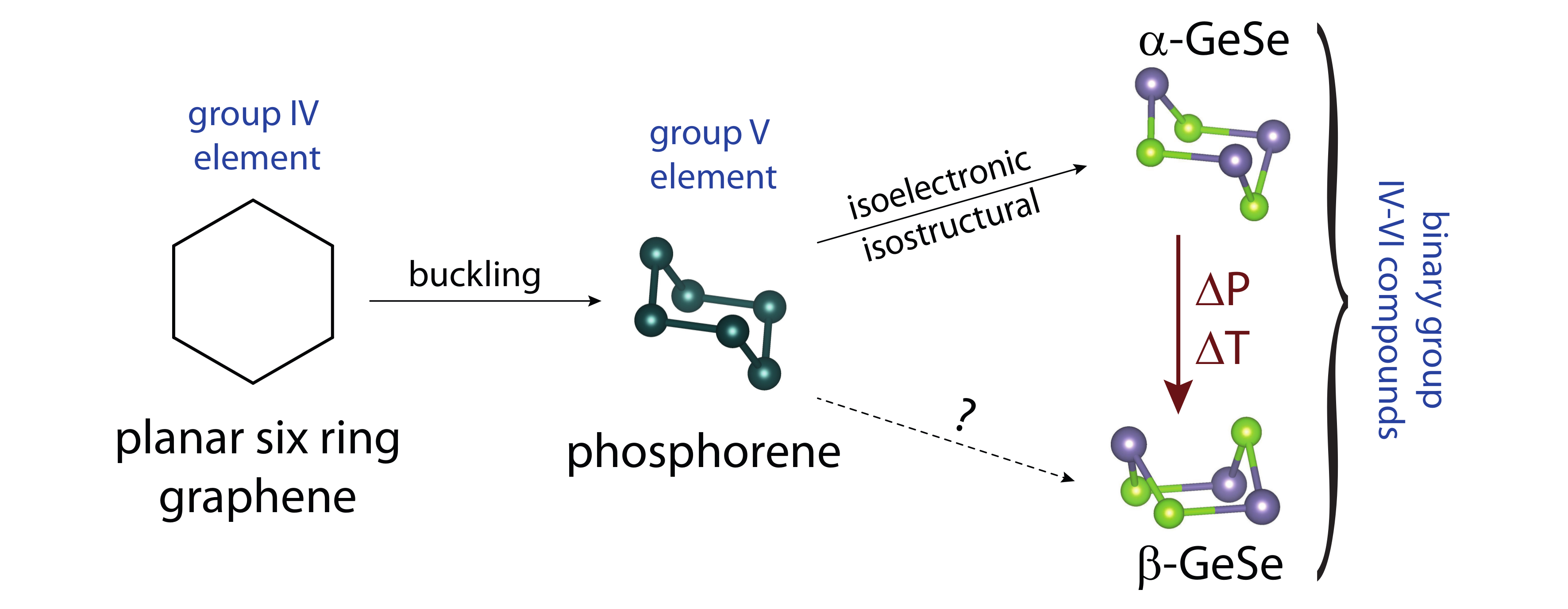
Two-dimensional materials such as graphene and transition metal dichalcogenides have shown good potential to act as the basis for future electronics technologies [1,2]. Similarly, black phosphorus is considered a promising candidate for many applications in electronics and optoelectronics, because, for a small band-gap semiconductor, it displays high mobility charge carriers and can be easily exfoliated [3,4,5]. The intrinsic lack of a band gap in graphene has been an obstacle for the construction of devices. Black phosphorus on the other hand has an intrinsic small band-gap, which can be tuned into a Dirac semimetal [6]. Black phosphorus is the thermodynamically stable phosphorus form, but it oxidizes when exposed to water and oxygen. Therefore, related layered semiconductors are of significant interest.
In this presentation, we report the electronic and structural properties of β-GeSe [7], a previously unreported polymorph of GeSe, with a unique crystal structure that displays strong two-dimensional structural features. β-GeSe is made at high pressure and temperature and is stable under ambient conditions. Here, we will compare it to its structural and electronic relatives α-GeSe and black phosphorus. The new β form of GeSe displays a boat configuration for its Ge-Se six-membered ring, while the previously known α form, and black phosphorus, display the more common chair configuration for their six-rings. Electronic structure calculations suggest that β-GeSe has a bulk band gap of Δ ≈ 0.5 eV, and, in its monolayer form, Δ ≈ 0.9 eV. These values fall between between those of α-GeSe and black phosphorus, making it a promising candidate for future applications.
[1] X. Ling, H. Wang, S. Huang, F. Xia, and M.S. Dresselhaus, Proc. Natl. Acad. Sci., 2015, 112, 4523.
[2] H. Liu, Y. Du, Y.Denga, and P.D. Ye, Chem. Soc. Rev., 2015, 44, 2732.
[3] L. Li, et al., Nat. Nanotech., 2014, 9, 372.
[4] L. Li, Yang, et al., Nat. Nanotech., 2016, 11, 593.
[5] J. Guo, H. Wang, F. von Rohr, W. Yi, Y. Zhou, Z. Wang, S. Cai, S. Zhang, X. Li, Y. Li, J. Liu, K. Yang, A. Li, S. Jiang, Q. Wu, T. Xiang, R. J. Cava, L. Sun, arXiv:1611.03330, 2016.
[6] J. Kim, S.S., et al., Science, 2015, 349, 723.
[7] F.O. von Rohr, H. Ji, F. A. Cevallos, T. Gao, N. P. Ong, and R. J. Cava, J. Am. Chem. Soc., 2017, 139, 2771.