Copper-Catalyzed Enantioselective Oxy and Amino-Alkynylations of Diazo Compounds
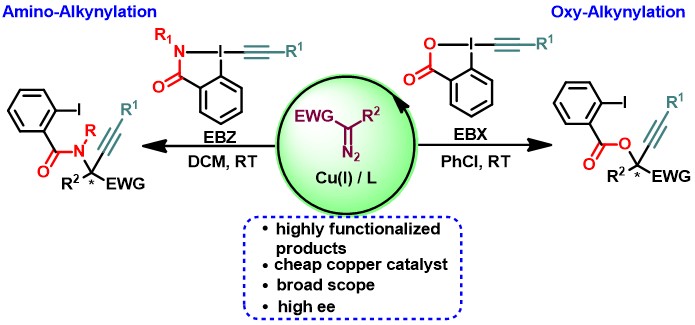
Alkynes are ubiquitous in both naturally occurring and synthetic organic compounds, which have found widespread applications in biology and materials sciences.1a Consequently, their synthesis is of fundamental interest in organic chemistry. One of the most often used methods for the synthesis of alkynes consists in the addition of acetylides on electrophilic positions in molecules. In contrast, the Umpolung-based alkynylations, the addition of alkynes onto nucleophiles, have been less investigated. In this context, hypervalent iodine reagents have been used for electrophilic alkynylations due to their exceptional reactivity.1b,1c However, alkynylations using hypervalent reagents generates one equivalent of an iodoarene as a side-product, leading to low atom economy. In 2016, our group developed an unprecedented copper-catalyzed oxy-alkynylation of diazo compounds based on the use of EthynylBenziodoXolones (EBX) reagents.2a This reaction is highly practical and proceeds under mild conditions for a broad range of substrates with high atom economy. Herein, we report the successful development of an enantioselective variation of this transformation, which represents the first example of asymmetric copper-catalyzed addition of both a nucleophile and an electrophile onto a carbenoid intermediate.2b The reaction exhibits broad scope towards both diazo compounds and hypervalent iodine reagents. The functional groups introduced during the transformation served as easy handles to access useful building blocks for synthetic and medicinal chemistry, such as α-hydroxy propargylic esters and vicinal diols, without loss of enantioselectivity. Furthermore, we have developed an enantioselective copper-catalyzed amino-alkynylation of diazo compounds using Ethynylbenziodazalone (EBZ) under mild conditions, giving access to unnatural amino acids.2c
[1] Diederich, F.; Stang, P. J.; Tykwinski, R. R. Acetylene Chemistry: Chemistry, Biology, and Material Science; Wiley-VCH: Weinheim, 2005; b) V. V. Zhdankin, P. J. Stang, Tetrahedron 1998, 54, 10927; b) Brand, J. P.; Waser, J. Chem. Soc. Rev. 2012, 41, 4165; c) Li, Y.; Hari, D. P.; Vita, M. V.; Waser, J. Angew. Chem., Int. Ed. 2016, 55, 4436.
[2] a) Hari, D. P.; Waser, J. J. Am. Chem. Soc. 2016, 138, 2190; b) Hari, D. P.; Waser, J. manuscript submitted; c) Hari, D. P.; Schouwey, L.; Barber, V.; Waser, J. manuscript in preparation.