Multicoordinated Precursors for Oxide Materials’ Synthesis and Other Applications
The specific arrangement and compartmentalization of metal ions are highly interesting for research in biology as well as in materials science. In order to program the coordination of two different metal ions, functional coordinating sections in ligands can be generated by design, leading to tweezer-type complexes with new properties stemming from the combination of the different metal ions.
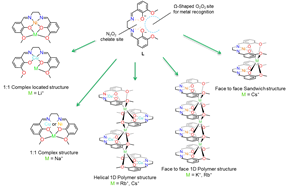
This project consists in the design of ligands with specific coordination sites to selectively bind different metal ions. The H2L ligand[1], a Schiff base derivative, has been synthetized possessing a central coordination site made of an imine-based N2O2 entity used to bind to copper(II) or nickel(II) upon deprotonation. This coordination prearranges the ligand into a Ω-shape, thus generating a second recognition site, O2O2, composed by two phenoxy and two methoxy functions.
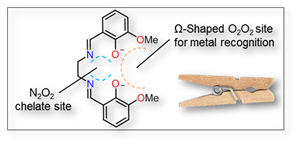
Structural differences influenced by alkali metal ions have been observed for copper(II) and nickel(II) compounds using single crystal X-ray diffraction.
The preorganization in these complexes allows also the synthesis of mixed oxide materials under mild conditions, controlling the stoichiometry of the final oxide material. The extreme synthesis conditions for classical solid state reactions (high pressure and/or temperature) can thus be avoided. The new mixed metal precursors are then simply combusted in air at low temperatures (ca. 300°C) to nanoscale oxide materials allowing to access new and better properties in their applications.
Further applications in mechanochemistry and for antimicrobial properties are currently investigated. One possible application is the one of optical sensing focused on the modification of the ligand for sensing applications.