Impact of degree of interaction and particle size on the efficiency of In2O3-based catalysts for CO2 hydrogenation to methanol
In order to reduce anthropogenic CO2 emissions, the identification of routes in which carbon dioxide can be utilized as a chemical feedstock is a highly sought goal in the scientific community. One attractive approach comprises its hydrogenation to methanol, which serves as a fuel and a starting material for the manufacture of a multitude of chemicals. So far, only one commercial process for CO2-based methanol production has been developed (Carbon Recycling International, 5 million liters in 2015),1 since most of the heterogeneous catalysts investigated suffer from limited selectivity, mainly due to the competitive reverse water gas shift reaction, and/or short lifetime. Recently, we have introduced indium oxide (9 wt.%) supported on zirconia as a highly selective and extraordinarily stable material for this transformation.2 In that study, we gathered evidence that the electronic interaction between the carrier and the active phase as well as the reaction conditions play a crucial role in the formation of selective active sites, i.e., surface oxygen vacancies. Here, we aim at achieving a deeper understanding of this catalyst to improve its methanol space time yield. In order to vary the distribution and degree of contact between In2O3 and ZrO2, we applied various preparation methods, i.e., wet impregnation, spray deposition, ball milling, hydrothermal synthesis, and co-precipitation. Preliminary tests under industrially-relevant temperature and pressure conditions showed that the first two protocols produce superior catalysts (Figure 1). Since XRD indicated that the particle size of In2O3 is smaller in these solids, we are currently producing additional materials with variable In content to tune the dimensions of the In2O3 crystallites. Characterization through a battery of state-of-the-art techniques will be coupled to the catalytic assessment to establish structure-performance relations and attain even more efficient materials.
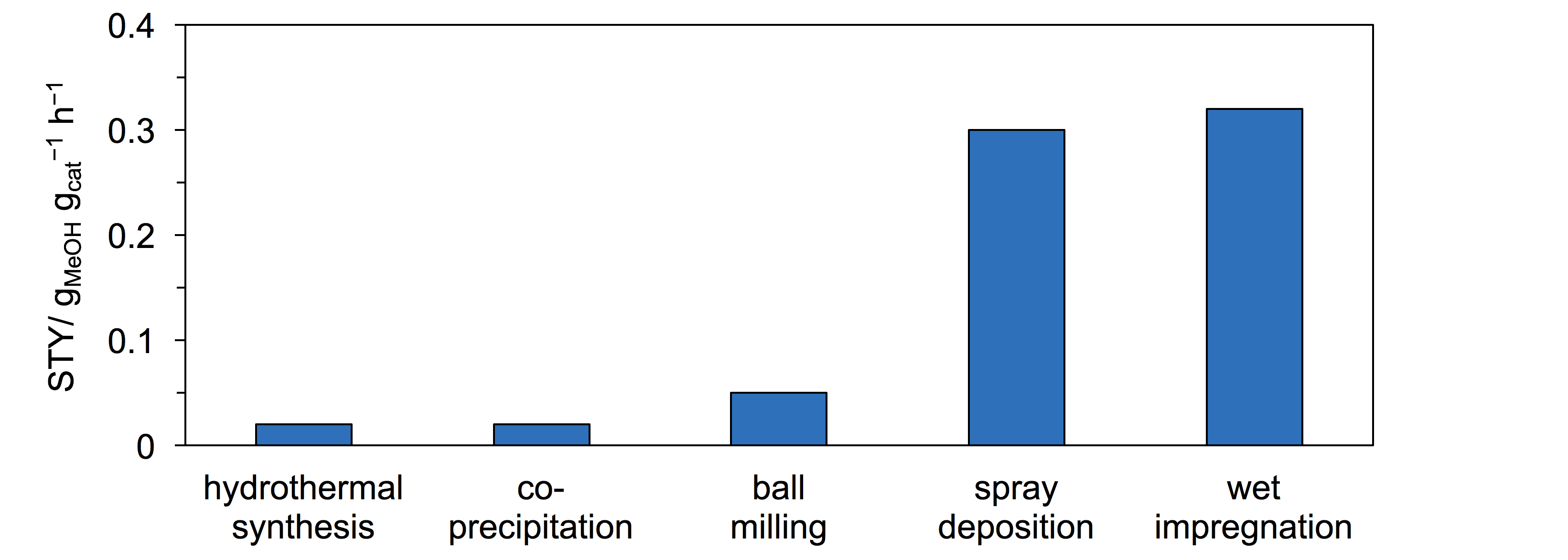
Figure 1 Space time yield (STY) of methanol in the direct hydrogenation of CO2 over In2O3-ZrO2 catalysts prepared by different synthesis methods.
[1] http://carbonrecycling.is/george-olah/2016/2/14/worlds-largest-co2-methanol-plant
[2] O. Martin, A. J. Martín, C. Mondelli, S. Mitchell, T. F. Segawa, R. Hauert, C. Drouilly, D. Curulla-Ferré, J. Pérez-Ramírez, Angew. Chem. Int. Ed. 2016, 55, 6261.