DNP Jelly
Dynamic Nuclear Polarization (DNP) is a hyperpolarization method that can deliver an increase in NMR signal intensity by up to 2 orders of magnitude, thereby permitting unprecedented atomic-level characterization of systems that were previously inaccessible such as cements or nanoparticles.[1] Dynamic nuclear polarization (DNP) experiments usually rely on the transfer of polarization from a radical containing matrix to a target of interest. Today, the matrix is either a glycerol-d8/D2O/H2O (6/3/1 v/v) mixture, so-called “DNP Juice"[2] or an organic liquid such as 1,1,2,2- tetrachloroethane[3] or ortho-terphenyl.[4] While these matrices work well, they present limitations in many cases, where solubility is partial, aggregation occurs at low temperatures, or if the substrate reacts with the matrix. Expanding the water based formulations range of matrices that can be used for DNP experiments is of considerable interest.
We present water based acrylamide gels as polarizing matrices for DNP. We observe that an increase in cross linker ratio combined with low concentration of biradical provides high enhancements comparable (ε = 200 for DNP Jelly) to the popular matrices available today, “DNP Juice”. The aqueous acrylamide gel allows the characterization of core-type CdTe-COOH quantum-dots using surface-enhanced NMR spectroscopy methods. The gels introduced here prevent aggregation while maintaining high enhancements, and Cd-113 spectra are acquired in minutes.
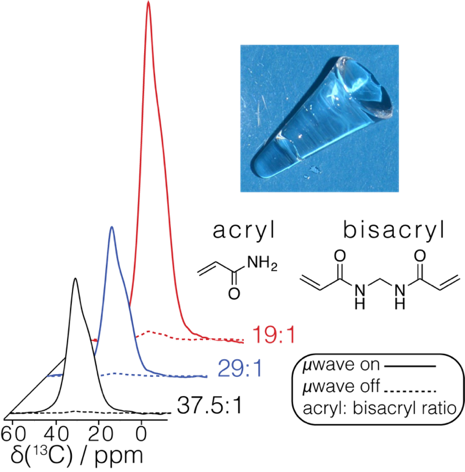
Figure 1. DNP enhancements in polyacrylamide water based gels as a function of pore size which decreases with the acryl: bisacryl ratio (given on the right).
[1] a) Rossini, A. J. ; Zagdoun, A. ; Lelli, M. ; Lesage, A. ; Copéret, C. ; Emsley, L. Acc. Chem. Res. 2013, (46), 1942-1951; b) Piveteau, L. ; Ong, T.-C. ; Rossini, A. J. ; Emsley, L. ; Copéret, C. ; Kovalenko, M. V. J. Am. Chem. Soc. 2015, (137), 13964-13971. [2] van der Wel, P. C. A.; Hu, K.-N.; Lewandowski, J.; Griffin, R. G., J. Am. Chem. Soc. 2006, (128) , 10840-10846. [3] Zagdoun, A.; Rossini, A. J.; Gajan, D.; Bourdolle, A.; Ouari, O.; Rosay, M.; Maas, W. E.; Tordo, P.; Lelli, M.; Emsley, L.; Lesage, A.; Coperet, C., Chem. Commun. 2012, (48), 654-656. [4] a) Ong, T.-C.; Mak-Jurkauskas, M. L.; Walish, J. J.; Michaelis, V. K.; Corzilius, B.; Smith, A. A.; Clausen, A. M.; Cheetham, J. C.; Swager, T. M.; Griffin R. G. J. Phys. Chem. B. 2013 (117), 3040-3046; b) Lelli, M.; Chaudhari, S. R.; Gajan, D.; Casano, G.; Rossini, A. J.; Ouari, O.; Tordo, P.; Lesage, A.; Emsley, L., J. Am. Chem. Soc. 2015 (137), 14558-14561.