Europium oxybromide catalysts for efficient bromine looping in natural gas valorization
The bromine-mediated upgrading of natural gas, comprising mainly methane and other light alkanes, is an attractive technology for decentralized production of value-added commodities.1,2 Its implementation primarily depends on the efficient recycle of stoichiometrically produced amounts of hydrogen bromide (HBr),1,2 which can be approached by the promising routes of catalytic HBr oxidation and intensified methane oxybromination (MOB).2,3 Nevertheless, the highly desirable low-temperature oxidation of HBr under a stoichiometric feed remains challenging since the extensive bromination of active catalysts (e.g. CeO2) typically leads to severe deactivation.2 In addition to stability and activity constraints, a high selectivity to methyl bromide (CH3Br) in MOB is also critical in the catalyst design.2,3 In this study, we demonstrate a novel europium oxybromide (EuOBr) catalyst exhibiting unprecedented performance in both HBr oxidation and MOB. In the former, EuOBr uniquely displays high bromine evolution activity at low-temperature and impeccable structural stability under practically-relevant conditions which are inaccessible by the state-of-the-art catalysts (Figure 1a,b). In the latter, a low propensity to combustion provides unparalleled selectivity to CH3Br (Figure 1c). By devising strategies to support EuOBr on an appropriate carrier in extrudate form, the utilization of europium can be enhanced by over an order of magnitude while preserving the exceptional catalytic properties (Figure 1d,e). This discovery paves the way for the implementation of sustainable bromine-mediated processes to harvest stranded natural gas for the production of readily transported chemicals and fuels.
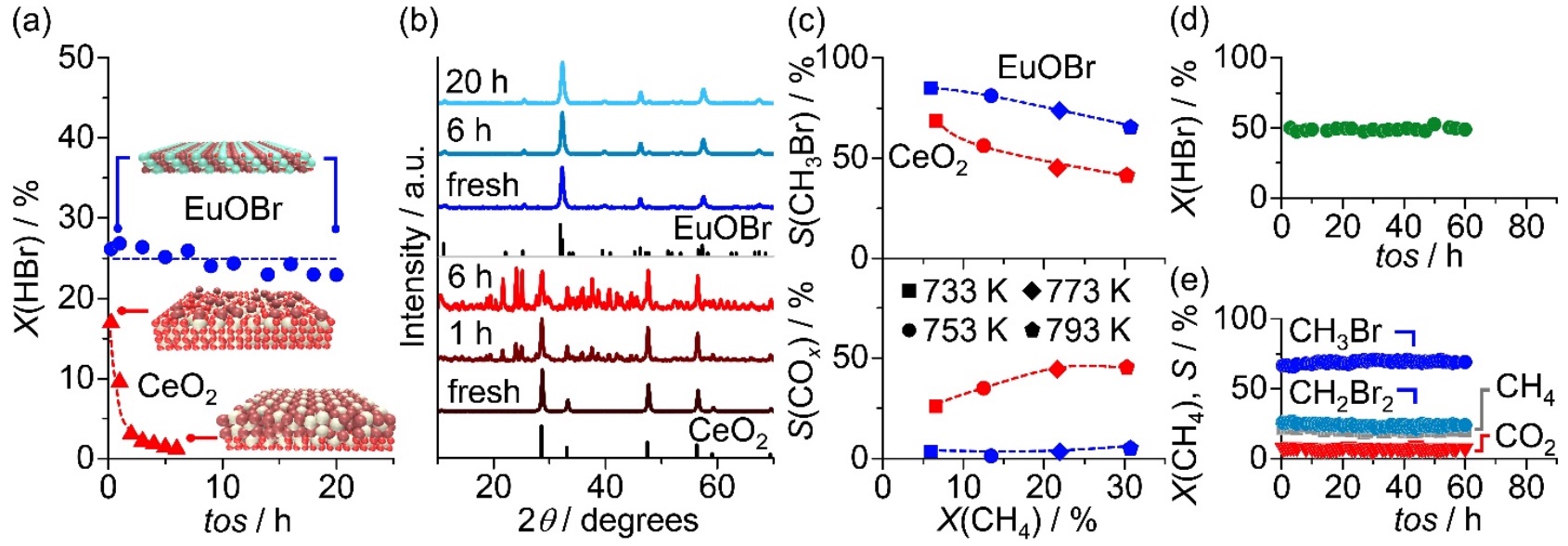
Figure 1 (a) HBr conversion, X(HBr), over EuOBr and CeO2 versus time-on-stream, tos. (b) XRD patterns of the catalysts recovered after various tos. (c) Selectivity to CH3Br, S(CH3Br), and carbon oxides, S(COx), versus CH4 conversion, X(CH4), in MOB over EuOBr and CeO2. (d) HBr oxidation over EuOBr/Al2O3 and (e) MOB over EuOBr/ZrO2 versus tos.
[1] E. McFarland, Science 2012, 338:341.
[2] R. Lin, A.P. Amrute, J. Pérez-Ramírez, Chem. Rev. 2017, 117:4182.
[3] V. Paunovic, G. Zichittella, A. P. Amrute, J. Pérez-Ramírez, Nat. Chem. 2016, 8:803.