FeVO4, CeVO4 and ErVO4-based catalysts for the selective catalytic reduction of NO with NH3
Efficient removal of NOx from exhaust gases of stationary sources is realized by reacting it with NH3 on a solid catalyst according to the standard selective catalytic reduction (SCR). Metal vanadate-based SCR catalysts exhibit promising advantages over traditional vanadia-based catalysts. Some MeVO4 were shown to be resistant to deactivation when supported on SiO2-WO3-TiO2 (TWS) and remained active after aging up to 750°C [1]. Nevertheless, it was recently shown that supported FeVO4 is not stable on TWS and decomposes into Fe2O3 and VOx species above 600°C [2, 3]. The intrinsic activity of the FeVO4-based SCR catalysts is rather low and is only enhanced by the thermal decomposition. In order to clarify the benefits of vanadates as SCR catalysts, we compared the activity and stability of FeVO4, CeVO4 and ErVO4 on a TWS support.
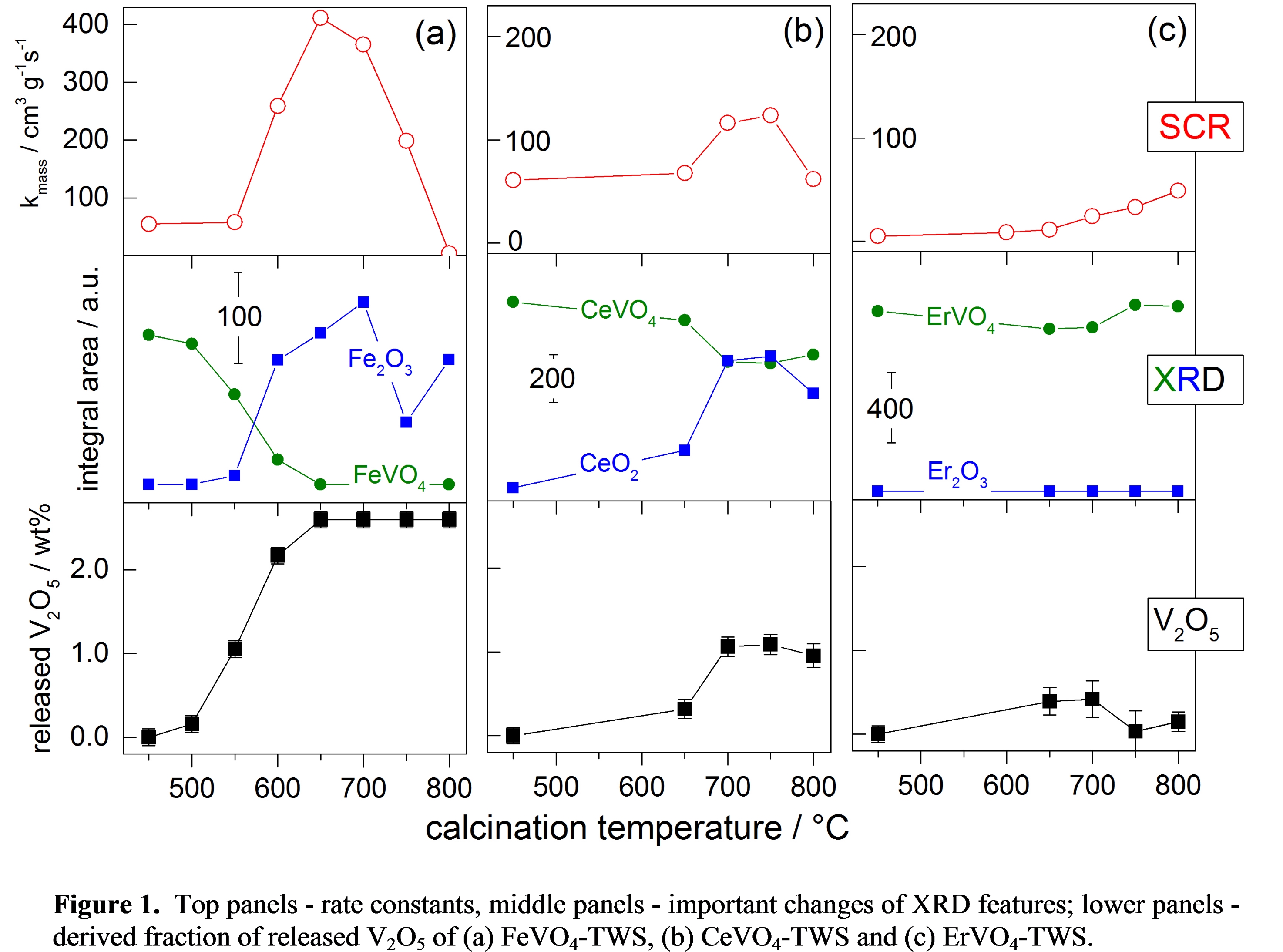
It was shown that an increase of calcination temperature activates all catalysts (increase in rate constant kmass, Figure 1) concurrent with the decrease of the metal vanadate peak integral from XRD. The decomposition of the metal vanadate gave rise to the corresponding metal oxides, but no traces of a new vanadium containing phase were observed. However, an estimation of released V2O5 could be derived from the loss of the metal vanadate phase (lower panels in Figure 1). The change of released V2O5 and kmass are strongly correlated. While FeVO4 decomposed completely, the maximum fraction of released V2O5 was 1.1 wt% for CeVO4-TWS and 0.4 wt% for ErVO4-TWS, hence CeVO4 and ErVO4 decomposed only partially. References with 0.5 and 1.0 wt% V2O5 on TWS showed very similar activity, indicating that the amount of released V2O5 is in fact responsible for the increase in activity. The existence of released V species from metal vanadates was proven using DRIFTS and DRUV, which revealed the predominant formation of extended VOx units on the surface of TWS. Summarizing, metal vanadate catalysts supported on TWS become active only after their decomposition into metal oxides and free VOx species.
[1] Casanova M., Schermanz K., Llorca J., Trovarelli A. Catal. Today, 2011 184, 227
[2] Marberger A., M. Elsener M., Ferri D., Sagar A., Schermanz K., Kröcher O. ACS Catal. 2015, 5, 4180 [3] Casanova M., Nodari L., Sagar A., Schermanz K., Trovarelli A. Appl. Catal. B. 2015, 176, 699