Design of a technical Mg-Al mixed oxide catalyst for the continuous manufacture of glycerol carbonate
Glycerol carbonate, obtained by the carbonylation of waste glycerol with urea, is an important bioderived chemical with applications as a solvent and an intermediate. Its preparation would ecologically and economically benefit from the availability of a heterogeneous catalyst, which contains cheap and abundant elements, has a scalable synthesis, is highly active and stable, retains its performance upon shaping into a technical form, and can be operated in continuous mode. In this work,1 we show that a mixed oxide of Mg and Al is a promising active phase for this reaction. The solid comprises widely available and non-toxic metals, is easily obtained through the thermal decomposition of a hydrotalcite-like material, and can almost match the product yield of state-of-the-art Zn-based catalysts2 (Figure 1a), while displaying an outstanding resistance against leaching, which causes the rapid dissolution of the latter. In-depth characterization uncovered that Lewis-basic centers are crucial to activate glycerol through dehydrogenation. Their concentration was maximized by optimizing the composition and calcination temperature of the precursor, thus reaching up to 60% glycerol carbonate yield (Figure 1a). Millimeter-sized extrudates showing comparable basic properties to the powder sample, a well-developed meso- and macroporosity, and high mechanical stability were obtained using a natural clay, bentonite, as a binder and thermally activating the hydrotalcite only after shaping. Upon testing in a continuous reactor under tuned conditions of temperature and pressure and in the presence of an aprotic solvent, the system showed the same glycerol yield as in the batch tests. During 100 h on stream, its activity decreased by 20% due to fouling (Figure 1b), but could be fully restored upon burning-off of the carbonaceous deposits. This work discloses the development of a green material that exhibits high efficacy in a sustainable transformation, highlighting key parameters for consideration in the design of an industrially-relevant chemocatalytic technology.
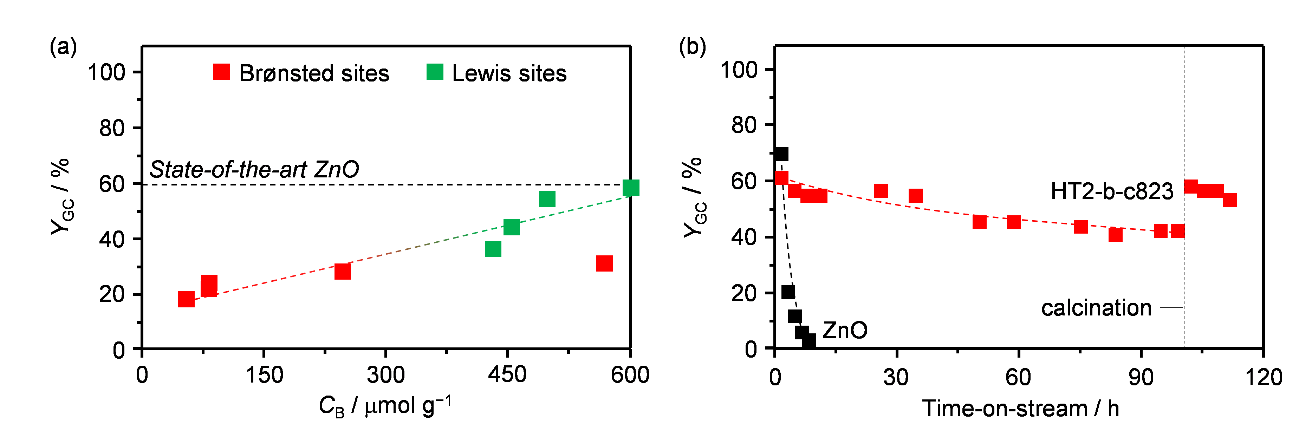
Figure 1 (a) Glycerol carbonate yield versus the concentration of basic sites for the hydrotalcite-derived catalysts and (b) evolution of the glycerol carbonate yield over 100 h and effect of regeneration by calcination on the performance.
[1] G. M. Lari, A. B. L. de Moura, L. Weimann, S. Mitchell, C. Mondelli, J. Pérez-Ramírez, J. Mater. Chem. A 2017, doi:10.1039/C7TA02061A.
[2] S. Fujita, Y. Yamanishi, M. Arai, J. Catal. 2013, 297, 137.