Mechanosensitive Membrane Probes and Beyond
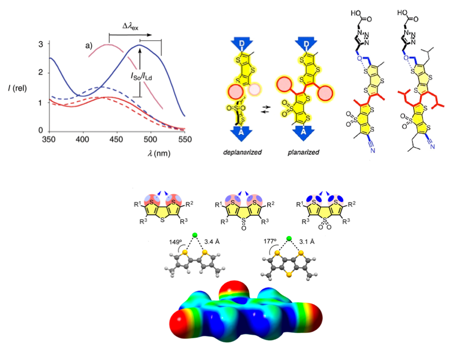
To understand the behaviour and the features of biological cellular membranes, mechanosensitive1 fluorescent “flipper” probes have been developed. Combining the chromophore polarization and ground state planarization is the key to determine and visualize the lateral organization.2 Improvement on chemical stability have been fully achieved through head group engineering using copper-catalyzed alkyne–azide cycloaddition (CuAAC) approach in a way to have probes ready for use in biology.3 Introducing “bulky” lateral chains on dithienothiophene (DTT) moyeties that affects the molecule’s twisted state, such as isobutyl groups, strongly hinders the planarization in the first excited state producing a probe that fails to respond to changes in membrane order.4 However the new more effective synthetic approach introduced, has been highly useful to develop DTT units as privileged motif to study anion transport5 and catalysis6 with chalcogen bonding. Expanding the length of the current flipper to “trimer” and “tetramer” units is now in progress with the aim to create a new generation of fluorescent membrane probes.
[1] Dal Molin, M.; Verolet, Q.; Soleimanpour S.; Matile, S. Chem. Eur. J. 2015, 21, 6012–6021.
[2] Dal Molin, M.; Verolet, Q.; Colom, A.; Letrun, R.; Derivery, E.; Gonzalez-Gaitan, M.; Vauthey, E.; Roux, A.; Sakai, N.; Matile, S. J. Am. Chem. Soc. 2015, 137, 568–571.
[3] Soleimanpour, S.; Colom, A.; Derivery, E.; Gonzalez-Gaitan, M.; Roux, A.; Sakai, N.; Matile, S. Chem. Commun. 2016, 52, 14450–14453.
[4] Macchione, M.; Chuard, N.; Sakai, N.; Matile, S. ChemPlusChem 2017, DOI: 10.1002/cplu.201600634.
[5] Benz, S.; Macchione, M.; Verolet, Q.; Mareda, J.; Sakai, N.; Matile, S. J. Am. Chem. Soc. 2016, 138, 9093–9096.
[6] Benz, S.; López-Andarias, J.; Mareda, J.; Sakai, N.; Matile, S. Angew. Chem. Int. Ed. 2017, 56, 812–815.