Semi hydrogenation of alkynes by a novel bimetallic rhodium(I) complex
Catalytic semi hydrogenation of alkynes to alkenes is an important process in both academia and industry. Developed in the 50’s, Lindlar’s catalyst (Pd/CaCO3/Pb(OAc)2/quinoline) still remains a privileged system for Z-selective semi hydrogenation reactions. However, the system utilizes a toxic lead additive. Further drawbacks are narrow substrate scope, over hydrogenation to alkenes, Z/E isomerization and double bond migration reactions. [1]
Herein, we report a new bimetallic complex 2 bearing a [Rh(I)]2-mojety. The stabilization of the rhodium-rhodium bond, while leaving two reactive rhodium centers, was achieved by a tailored ligand with olefin/alkyne sites to anchor the two metal centers. The complex 2 was shown to be active in semi hydrogenation of alkynes under very mild conditions. In order to optimize the stability and activity of the complex, high throughput ligand- and substrate screening was performed, leading to a catalyst system able to perform semi hydrogenation reactions with up to 94 % alkene selectivity.
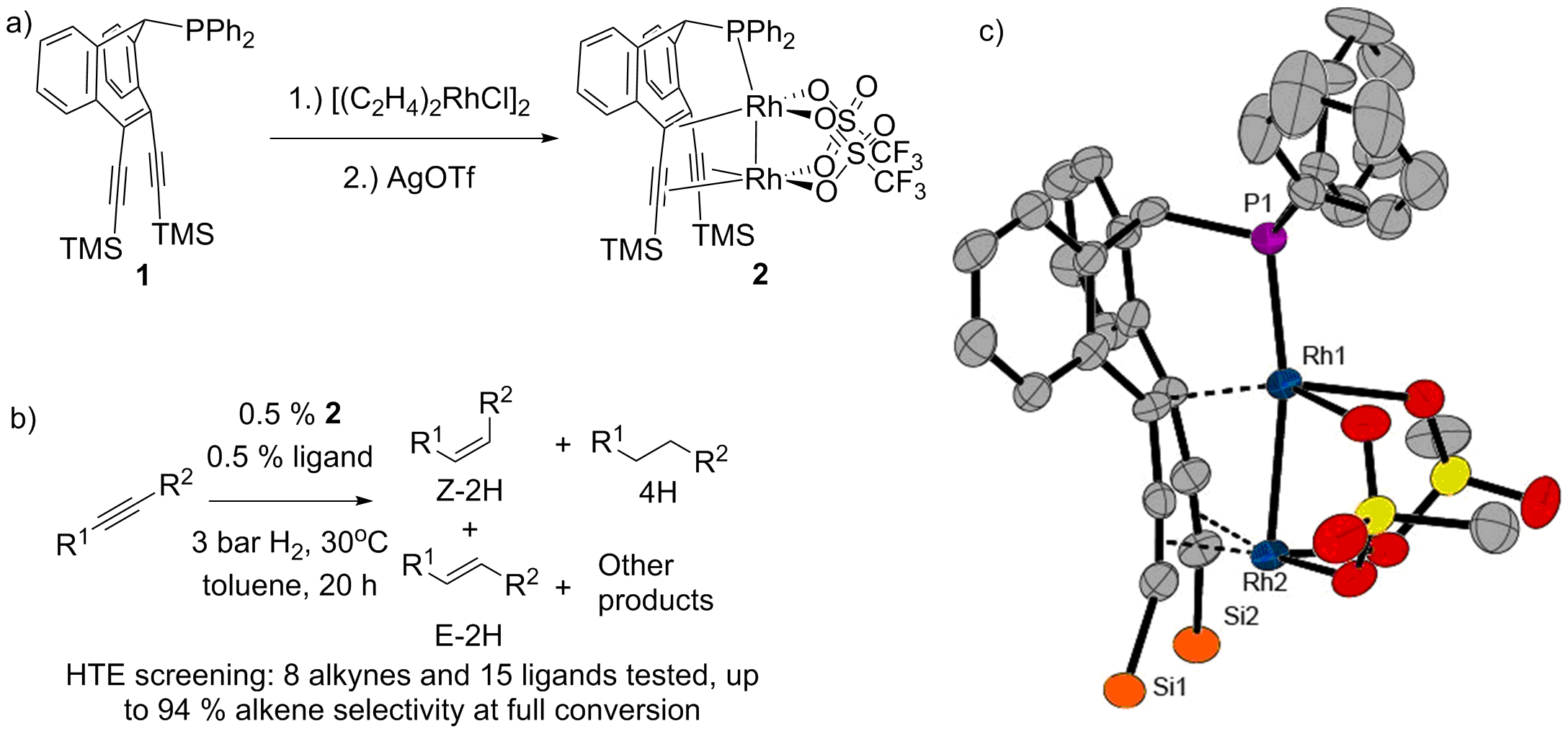
Figure 1. a) Synthesis of a dirhodium complex 2, b) Conditions for semi hydrogenation reactions, c) crystal structure of 2. Methyl groups and fluorine atoms omitted for clarity.
[1] A. Fedorov, H. Liu, H. Lo, C. Copéret, J. Am. Chem. Soc. 2016, 138, 16502.