Hydrodeoxygenation of bio-derived phenols over Pt Nanoparticle/Ionic Liquid Catalytic System under mild conditions
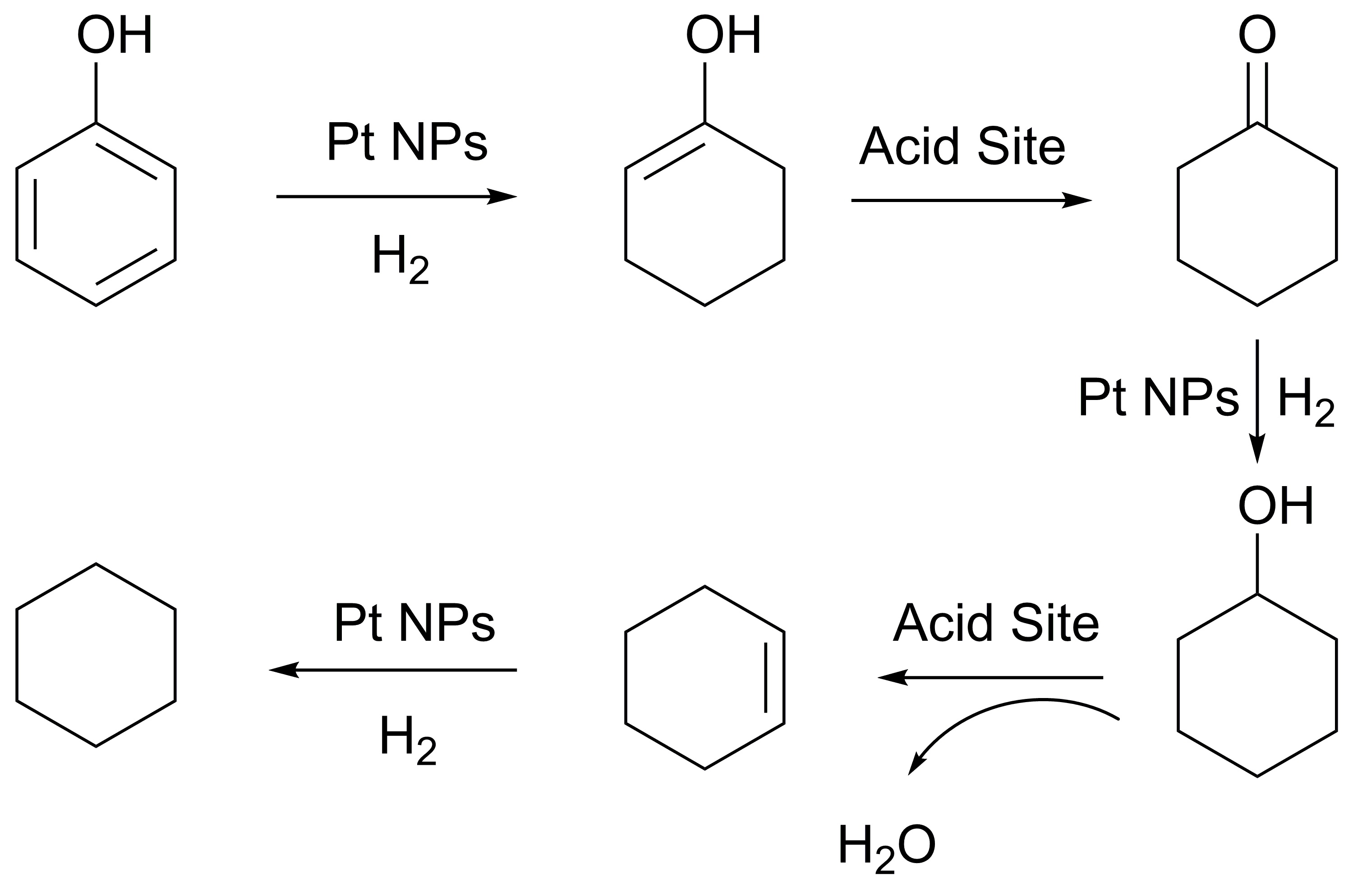
Due to the lower heating value and thermal instability, bio-oil cannot be used directly to replace diesel.1 To upgrade bio-oil through hydrodeoxygenation (HDO), appropriate catalysts and solvent systems would be indispensable.2 In our study, uniformly stabilized platinum metal nanoparticles (NPs) in ionic liquids (ILs) formed an efficiency catalyst system that performs the HDO of phenol, one of the most abundant components in bio-oil, under mild conditions (60 °C, 1.0 MPa H2). Pt NPs with a mean diameter of 3.4 nm (Fig. 1) in ILs with full conversion and maximum 94% selectivity to cyclohexane and cyclohexene, is the best combination. A possible reaction pathway of the HDO of phenol is proposed (Scheme 1). Further optimization of the reaction conditions including temperature, pressure, time and the preparation of the NPs were also carried out.

To figure out the C-O cleavage behavior between aromatic units, dimeric model compound diphenyl ether with C-O bonds (4-O-5) were investigated, meanwhile, other substituted phenols with hydroxy groups rather than methoxy groups, such as catechol, guaiacol and resorcinol were studied as well. In most cases afforded derivatives of cyclohexanes in good yields. Our findings indicate that the presented NPs/ILs catalytic systems are more active towards the removal of hydroxy groups rather than methoxy groups.
[1] Majid Saidi et al. Energy Environmental Science, 2014, 7, 103–129.
[2] Jiayue He et al. Journal of Catalysis, 2014, 309, 362–375.