Transforming Olefins into Dinucleophiles
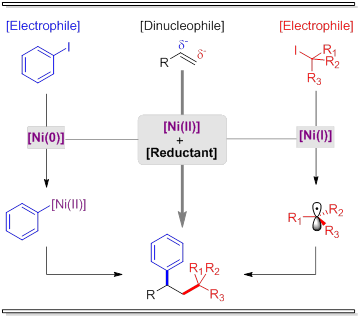
Addition of two hydrocarbon derivatives across π-systems has been traditionally carried out using formally a nucleophile and an electrophile as reaction partners[1]. The multiple bond is thus conceptually considered as a (+/-)-dipole, which justifies the extensive use of electronic biased substrates.[2] Alkylative procedures that can be applied to a wide range of substrates independently of their intrinsic nature are still in high demand. Here, we present the first example of an intermolecular, three-component reductive dicarbofunctionalization of olefins with aryl iodides and alkyl iodides as reaction partners.[3] In contrast to previous methods[4], the reaction tolerates a wide range of olefins and the carbon sources do not require any additional functionalization. Interestingly, this process is characterized by the present of two different nickel cycles interconnected by the action of both, the unsaturated partner and the reductant.
[1] a) E. Negishi, Z. Huang, G. Wang, S. Mohan, C. Wang, H. Hattori, Acc. Chem. Res. 2008, 41, 1474; b) A. B. Flynn, W. W. Ogilvie, Chem. Rev. 2007, 107, 4698; c) M. Ihara, K. Fukumoto, Angew. Chem. Int. Ed. 1993, 32, 1010; d) M. J. Chapdelaine, M. Hulce Org. React. 1990, 38, 225.
[2] For our previous contributions see: a) Z. Li, A. García-Domínguez, C. Nevado, Angew. Chem. Int. Ed. 2016, 55, 6938; b) Z. Li, A. García-Domínguez, C. Nevado, J. Am. Chem. Soc. 2015, 137, 11610.
[3] A. García Domínguez, Z. Li, C. Nevado J. Am. Chem. Soc. 2017, DOI: 10.1021/jacs.7b03195.
[4] For Ni-catalyzed methods see: a) J.-W. Gu, Q.-Q. Min, L.-C. Yu, X. Zhang Angew. Chem. Int. Ed. 2016, 55, 12270; b) T. Qin, J. Cornella, C. Li, L. R. Malins, J. T. Edwards, S. Kawamura, B. D. Maxwell, M. D. Eastgate, P. S. Baran, Science 2016, 352, 801; c) J. Terao, F. Bando, N. Kambe, Chem. Commun. 2009, 7336.