Heptacoordinate Co(II) complex: a new architecture for photochemical hydrogen production
Hydrogen is a promising potential clean source of energy which can be produced using abundant and renewable resources (e.g. water and sunlight). Water reduction catalysts are usually based on non-precious metals among which cobalt has been the most studied because of its abundance and low price. However the development of highly active and stable catalysts that can operate in purely aqueous solutions still remains a great challenge.[1]
The most active molecular cobalt catalysts reported in literature present a common architecture: a tetra- or pentapyridyl ligand inducing a distorted octahedral geometry, with the remaining coordination sites occupied by labile ligands (e.g. water).[2,3]
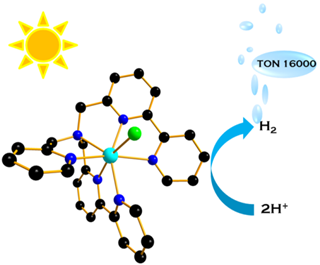
To further investigate the effect of coordination geometry on the catalytic activity, we designed a new ligand with six coordination sites that lead to the formation of an unusual heptacoordinate Co(II) complex. Under visible light irradiation in water, this complex can efficiently catalyze the production of H2 with a turnover number (TON) > 16000 mol H2 (mol cat)-1 and nearly 90% of H2 evolved within the first hour of irradiation, i.e. a turnover frequency (TOF) > 4000 mol H2 (mol cat)-1 h-1. [4]
These results suggest that heptacoordinate cobalt complexes, never used so far in the field of light-driven hydrogen evolution, represent a promising alternative platform for the development of highly active and stable photocatalysts.
[1] Artero, V.; Chavarot-Kerlidou, M.; Fontecave, M. Angew. Chem. Int. Ed. 2011, 50, 7238-7266.
[2] Bachmann, C.; Guttentag, M.; Spingler, B.; Alberto, R. Inorg. Chem. 2013, 52, 6055-6061.
[3] Singh, W. M.; Baine, T.; Kudo, S.; Tian, S. L.; Ma, X. A. N.; Zhou, H. Y.; DeYonker, N. J.; Pham, T. C.; Bollinger, J. C.; Baker, D. L.; Yan, B.; Webster, C. E.; Zhao, X. Angew. Chem. Int. Ed. 2012, 51, 5941-5944.
[4] Lucarini, F.; Pastore, M.; Vasylevskyi, S.; Varisco, M.; Solari, E.; Crochet, A.; Fromm, M.K.; Zobi, F.; Ruggi, A. Chem. Eur. J. 2017, DOI: 10.1002/chem.201701427.