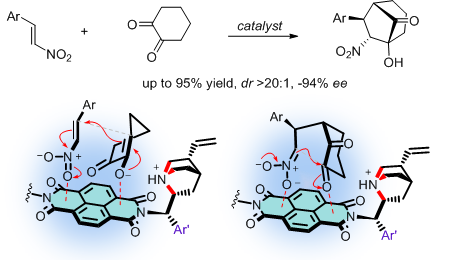
Asymmetric Anion-π Catalysis: Diastereospecific Michael/Henry Reactions for Bicyclic Prodcuts with Quarternary Chiral Centers on NDI Surfaces
The functional relevance of anion-π interactions has been integrated into viarious systems including anion recongnition, binding, transport and catalysis.1 The general ideal to use anion-π interactions in catalysis is to stabilize negatively charged intermediates and transition states on π-acidic surfaces. The concept has been explicitly proved validate2 first in 2013 and later on realized also for complex reaction systems including asymmetric enamine activation,3 iminium cascade processes4 and the first anion-π enzyme.5 As a new step forward, we are now extending anion-π catalysis to a more complicated cascade system to prepare bicyclic compounds with four stereogenic centers including one quaternary carbon center from achiral substrates. Hybridization of cinchona alcaloids with naphthalenediimides (NDI) affords a new anion-π cinchona fusion catalyst which results in much improved diastereoselectivity and enantioselectivity compared to previous catalysts and controls. Moreover, the cascade transformation was also realized by artificial anion-π enzyme in neutral water. Evidence in support of the relevance of anion-π interactions in catalyzing the cascade process include increasing stereoselectivities and velocities in the presence of π-acidic surfaces and inhibition with anions in order of NO3–, Br–, BF4–, PF6–.
[1] Zhao, Y.; Cotelle, Y.; Sakai, N.; Matile, S. J. Am. Chem. Soc. 2016,138, 4270. [2] Zhao,Y.; Domoto, Y.; Orentas, E.; Beuchat, C.;Emery, D.; Mareda, J.; Sakai, N.; Matile, S. Angew. Chem. Int. Ed. 2013, 52, 9940. [3] Zhao, Y.; Cotelle, Y.; Avestro, A.-J.; Sakai, N.; Matile, S. J. Am. Chem. Soc. 2015, 137, 11582. [4] Liu, L.; Cotelle, Y.; Avestro, A.-J.; Sakai, N.; Matile, S. J. Am. Chem. Soc. 2016, 138, 7876. [5] Cottelle, Y.; Lebrun, V.; Sakai, N.; Ward, T. R.; Matile, S. ACS. Cent. Sci. 2016, 2, 388.