Ketenimines from Isocyanides and Allyl Carbonates: Palladium-Catalyzed Synthesis of β,γ-Unsaturated Amides and Tetrazoles
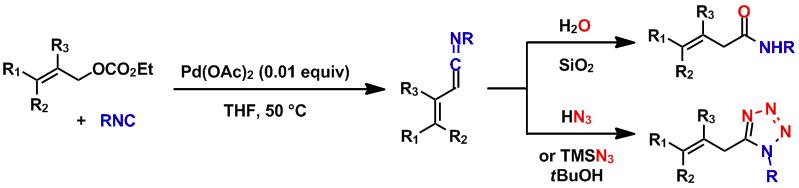
The reaction of allyl ethyl carbonates with isocyanides in the presence of a catalytic amount of Pd(OAc)2 provided ketenimines through β-hydride elimination of the allyl imidoylpalladium intermediates. The insertion of the isocyanide into the π-allyl Pd complex proceeded via an unusual η1-allyl Pd species. The resulting ketenimines were hydrolyzed to β,γ-unsaturated carboxamides during purification by flash column chromatography on silica gel or converted in situ into 1,5-disubstituted tetrazoles by [3+2] cycloaddition with hydrazoic acid or trimethylsilyl azide.
[1] Dr. Guanyinsheng Qiu†, Mathias Mamboury†, Dr. Qian Wang and Prof. Dr. Jieping Zhu, Angew. Chem. Int. Ed. 2016, 55 ,15377 –15381
† These authors contributed equally.