A Bidentate Mo(0) Isocyanide Complex is a Powerful Photoredox Catalyst
Photoredox catalysis has received significant attention in recent years, providing mild reaction conditions for a wide range of photoreactions in organic syntheses.[1] However, this class of reactions is often limited by the reducing power of the photocatalysts. To further improve the substrate scope, the development of stable photocatalysts capable of performing more demanding chemical transformations is desirable.
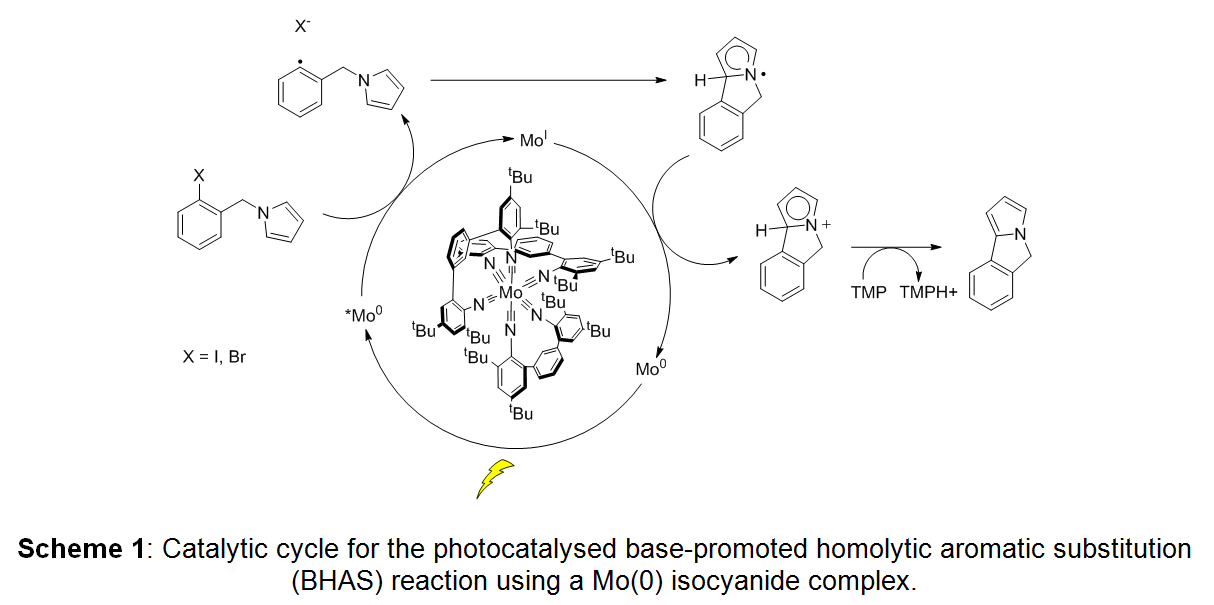
We demonstrated that bidentate isocyanide complexes of molybdenum(0) can act as strong photoreductants.[2,3] With an excited state oxidation potential of -1.9 V vs. SCE, the Mo(0) complex in Scheme 1 is 0.2 V more reducing than the widely used [Ir(ppy)3] complex.
We present the photophysical properties of this Mo(0) complex and demonstrate its application in photoredox catalysis. A specific example is the photocatalytic base-promoted homolytic aromatic substitution (BHAS) reaction shown in Scheme 1.
[1] Megan Shaw, Jack Twilton, Dave MacMillan, J. Org. Chem., 2016, 81, 6898–6926
[2] Laura A. Büldt, Xingwei Guo, Alessandro Prescimone, Oliver S. Wenger, Angew. Chem. Int. Ed., 2016, 55, 11247-11250
[3] Laura A. Büldt, Oliver S. Wenger, Angew. Chem. Int. Ed., 2017, 56, 5676-5682