Enantioselective Total Synthesis of (+)-Peganumine A
(+)-Peganumine A, a dimeric tetrahydro-β-carboline alkaloid, was isolated in 2014 from Peganum harmala L. by Li, Hua and coworkers.1 This unprecedented octacyclic compound contains two quaternary stereocenters embedded in a unique 3,9-diazatetracyclo[6.5.2.0.0]pentadec-2-one scaffold. This molecule shows low µM cytotoxicity against various cancer cell lines.
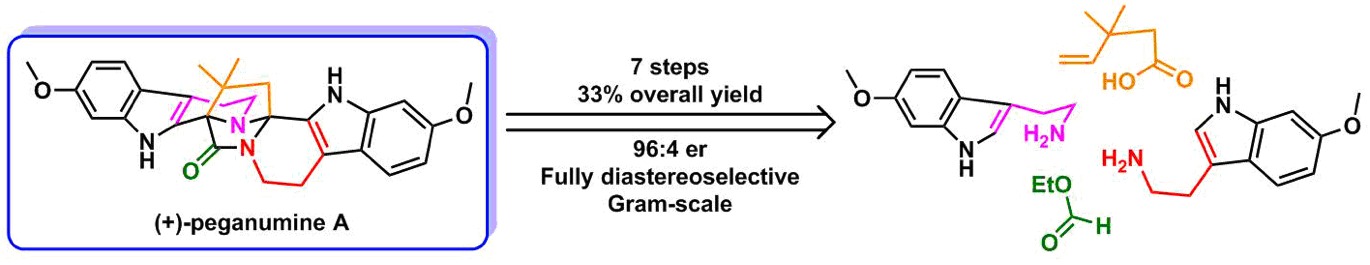
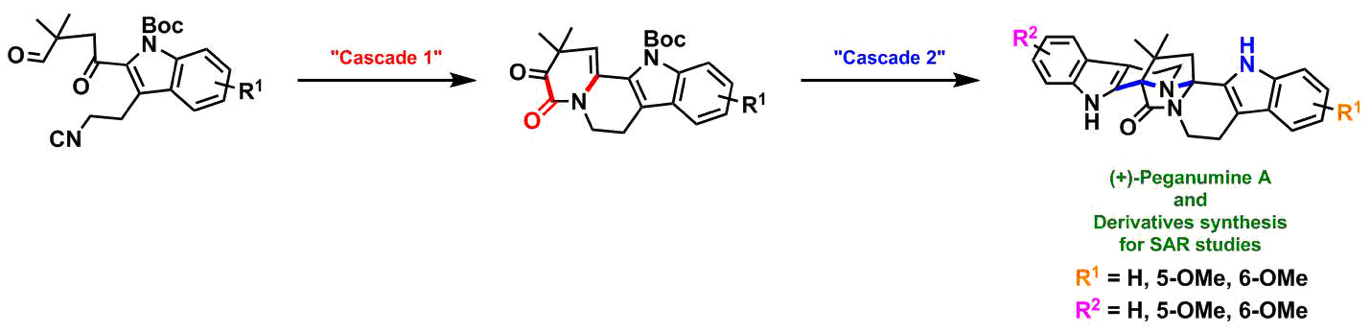
The intriguing molecular architecture in conjunction with its significant bioactivity and extremely low isolation yield prompted us to undertake the total synthesis of peganumine A. Starting from simple commercially available materials, we used two key cascade reactions to reach the final target:
- “Cascade 1”: A one-pot construction of an indolo[2,3-a]quinolizine tetracyclic skeleton from an ω-isocyano-γ-oxo-aldehyde via a sequence of an unprecedented C-C bond forming lactamization and a transannular condensation.
- “Cascade 2”: A one-pot process merging two achiral building blocks into the final enantioenriched octacyclic structure via a sequence of asymmetric Pictet-Spengler reaction followed by an acid-catalyzed transannular cyclization. This domino process created two quaternary stereocenters with concurrent formation of two spirocycles and the 2,7-diazabicyclo[2.2.1]heptan-3-one unit with control of both the absolute and the relative stereochemistry.
In a single pass, we have synthesized gram-quantity of (+)-peganumine A in 7 steps and 33% overall yield.2 This convergent route allowed us to synthesize the derivatives of peganumine A for future structure-activity relationship studies.
[1] K.-B. Wang, Y.-T. Di, Y. Bao, C.-M. Yuan, G. Chen, D.-H. Li, J. Bai, H.-P. He, X.-J. Hao, Y.-H. Pei, Y.-K. Jing, Z.-L. Li, H.-M. Hua, Org. Lett. 2014, 16, 4028-4031.
[2] C. Piemontesi, Q. Wang, J. Zhu, J. Am. Chem. Soc. 2016, 138, 11148-11151.