Surface Functionalization of Metal Nanoparticles and the Effect of the Polymer Topology on the Colloidal Stability
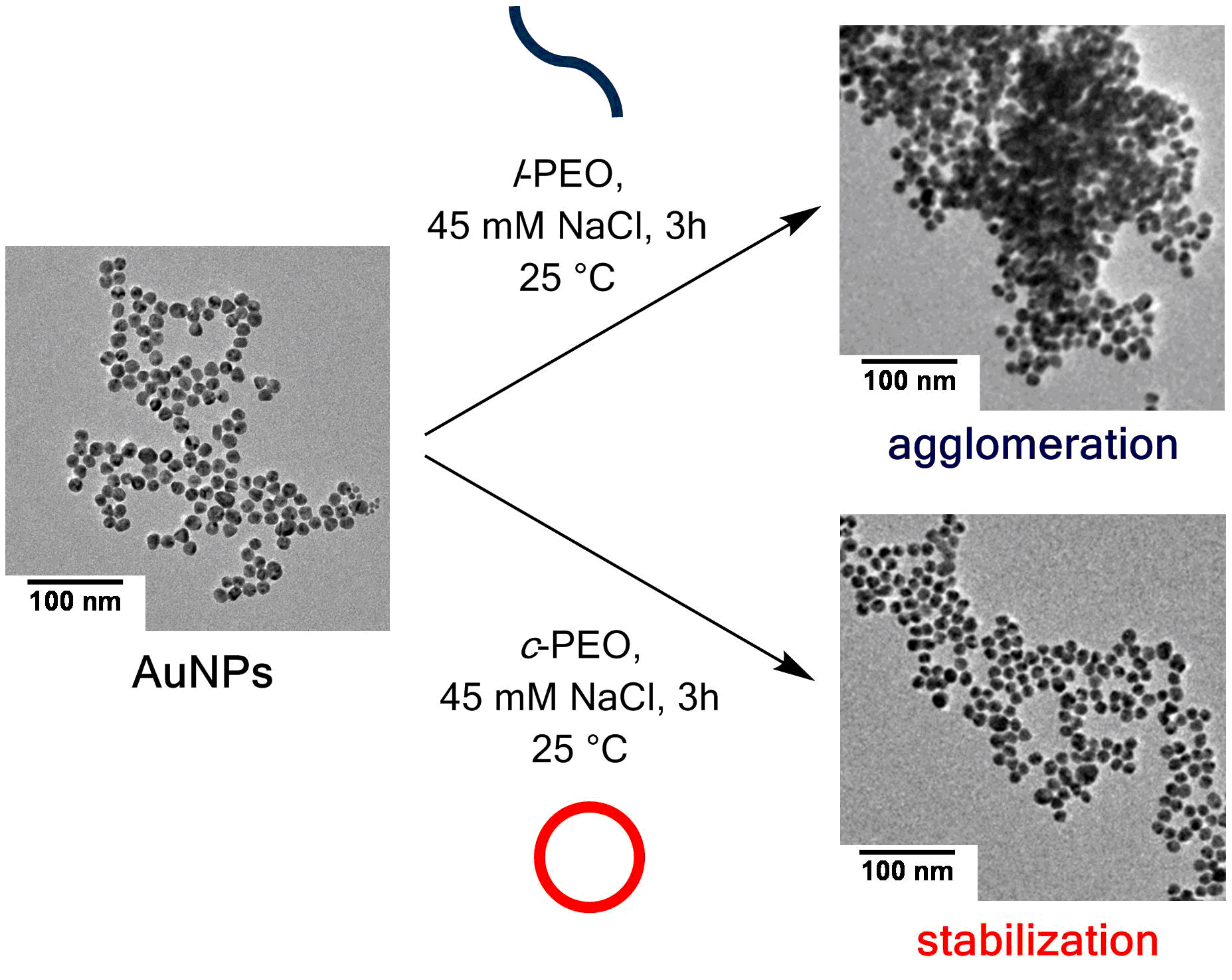
Metal nanoparticles exhibit an array of properties which have enabled their use in various scientific disciplines. However, the preservation of the nanoparticle stability towards agglomeration or dissolution remains a major drawback since those phenomena alter the size- and shape-dependent properties of the nanostructures. The key to the enhancement of the stability is the tuning of the surface properties of the nanoparticles, and various approaches have already been conducted in order to achieve this goal. For instance, the surface of metal nanoparticles has already been functionalized with various inorganic, organic or hybrid inorganic-organic shells in order to tailor them for their specific target applications. This strategy enabled the preparation of non-conducting metal nanoparticles suitable as dielectric materials [1] or core-shell particles rendered hydrophobic through surface-initiated radical polymerization beneficial for their dispersion in different organic solvents [2]. Although a variety of polymers have already been grown or attached onto the nanoparticle surface or have simply been applied as additional stabilizer into the colloidal dispersion, the effect of the polymer topology on the enhancement of the nanoparticle stability has not yet been subjected to thorough investigation. Previous work featured that micelles composed of cyclic ABA triblock copolymers exhibit enhanced stabilities towards salt-induced agglomeration compared to the micelles of their corresponding linear counterparts.[3] Hence, a key highlight of this work features the synthesis and stabilization of metal nanoparticles (gold and silver) by polyethylene oxide (PEO) where substantial changes in the average nanoparticle size and stability under saline conditions were observed through the variation of the polymer topology. Overall, the average particle size of the metal nanoparticles is reduced in the presence of cyclic PEO (c-PEO) and the resistance of the nanoparticles towards agglomeration is extended over a larger concentration of NaCl (≤ 180 mM) which is in direct contrast to the nanoparticles stabilized by linear PEO (l-PEO) where agglomeration occurs at NaCl concentrations as low as 45 mM.
[1] Jose Enrico Quijano Quinsaat, Frank Alain Nüesch, Heinrich Hofmann, Dorina Maria Opris, RSC Advances, 2013, 3, 6964-6971.
[2] Jose Enrico Quijano Quinsaat, Frank Alain Nüesch, Heinrich Hofmann, Dorina Maria Opris, RSC Advances, 2016, 6, 44254-44260.
[3] Satoshi Honda, Takuya Yamamoto, Yasuyuki Tezuka, Nature Communications, 2013, 4, 1574.