Labeling, mutations and truncated constructs: approach to solving the structure of the CPEB3 ribozyme through NMR spectroscopy
Ribozymes are RNA molecules capable of catalyzing specific chemical reactions. Several small ribozymes were recently discovered in mammals1. The cytoplasmic element binding protein 3 (CPEB3) ribozyme is a self-cleaving ribozyme that was found to be highly conserved in the mammalian genome1. Characterization of the CPEB3 ribozyme showed that it has similar properties as the hepatitis delta virus (HDV) ribozyme, including the fold and catalytic mechanism1,2,3. Unlike some self-cleaving ribozymes such as the hammerhead, hairpin and VS ribozymes where high concentrations of monovalent metal ions can promote catalysis, Mg2+ is required by the CPEB3 and HDV ribozymes4. Both ribozymes fold in a sophisticated nested double pseudoknot motif. According our previously work3, the CPEB3 ribozyme is significantly compacted with monovalent ions alone but the active fold is only achieved after addition of millimolar amounts of Mg2+. NMR studies showed the formation of a GU wobble pair with addition of Mg2+, which may correspond to the GU wobble that is an essential part of the active site in the HDV ribozyme5.
Here, we aim to solve the solution structure of the CPEB3 ribozyme through NMR spectroscopy. The assignment of the 1H-1H NOESY spectra of the full length ribozyme is a challenge due to the limited building blocks of RNA as well as the length of the CPEB3 ribozyme of 67 residues. In order to alleviate the spectral overlap, various labeled nucleotides were incorporated through in vitro transcription including partially deuterated, fully deuterated and 13C, 15N-labeled nucleotides.
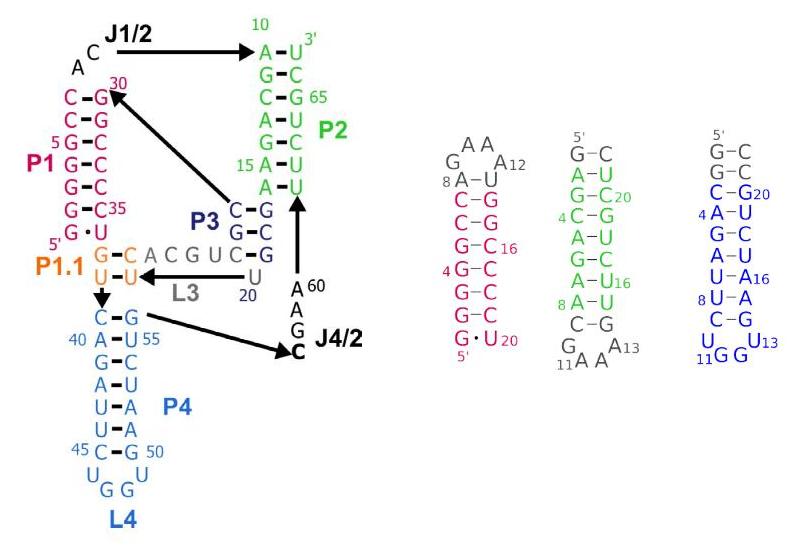
Figure 1. Secondary structure of the chimp CPEB3 ribozyme with auxiliary constructs of domains P1 (red), P2 (green) and P4 (blue)
Financial support by the Swiss National Science Foundation is gratefully acknowledged.
[1]Salehi-Ashtiani, K., Lupták, A., Litovchick, A. and J.W. Szostak. Science, 2006. 313 (5794): p. 1788-1792
[2]Chadalavada D.M., Gratton, E.A. and P.C. Bevilacqua. Biochemistry, 2010. 49(25): 5321-5330.
[3]Skilandat, M. Rowinska-Zyrek, M. and R.K.O. Sigel. RNA, 2016. 22(5): 750-763
[4]Murray, J.B., Seyhan A.A., Walter, N.G., Burke J.M. and W.G. Scott. Chem. Biol., 1998. 5:587-595.
[5]Chen, J.H., Yajima, R., Chadalavada, D.M., Chase, E., Bevilacqua, P.C. and B.L. Golden. Biochemistry, 2010 49:6508-6518.