Iron(II) Complexes of Chiral, C2-Symmetric, P- or C-Stereogenic PNP Pincer Ligands and their Application in the H2 Hydrogenation of Ketones
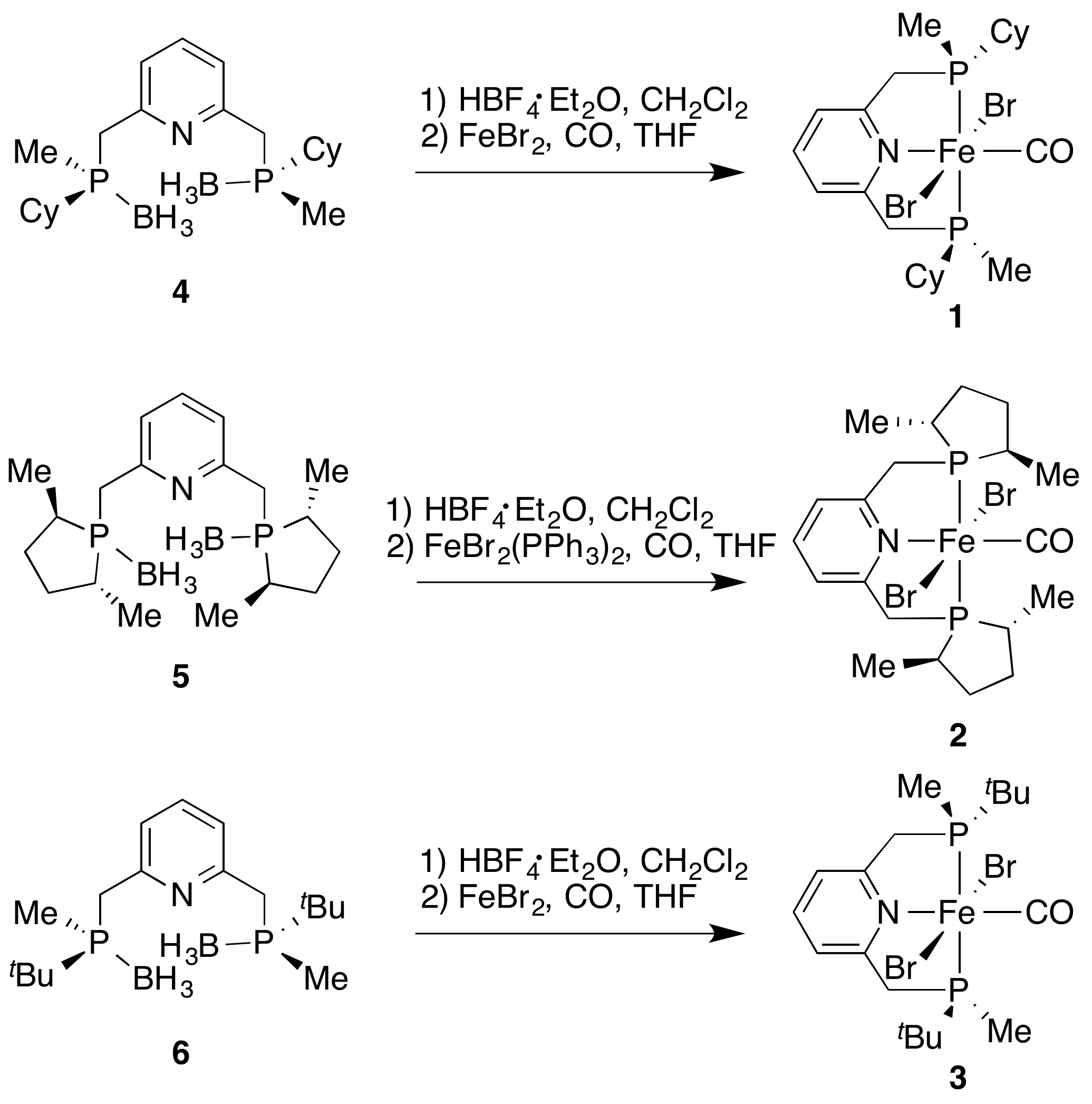
Chiral pincer ligands for iron(II) have been recently developed and applied in the direct H2 hydrogenation of ketones.1,2 We report here the preparation and characterization of iron(II) dibromocarbonyl complexes 1, 2, and 3 of the type [FeBr2(CO)(L)], where L is one of the tridentate, P- or C-stereogenic, C2-symmetric PNP pincer ligands (SP,SP)-2,6-bis((cyclohexyl(methyl)phosphanyl) methyl)pyridine (4), 2,6-bis(((2R,5R)-2,5-dimethylphospholan-1-yl)methyl)pyridine (5), or (SP,SP)-2,6-bis( (tert-butyl(methyl)phosphanyl)methyl) pyridine (6).
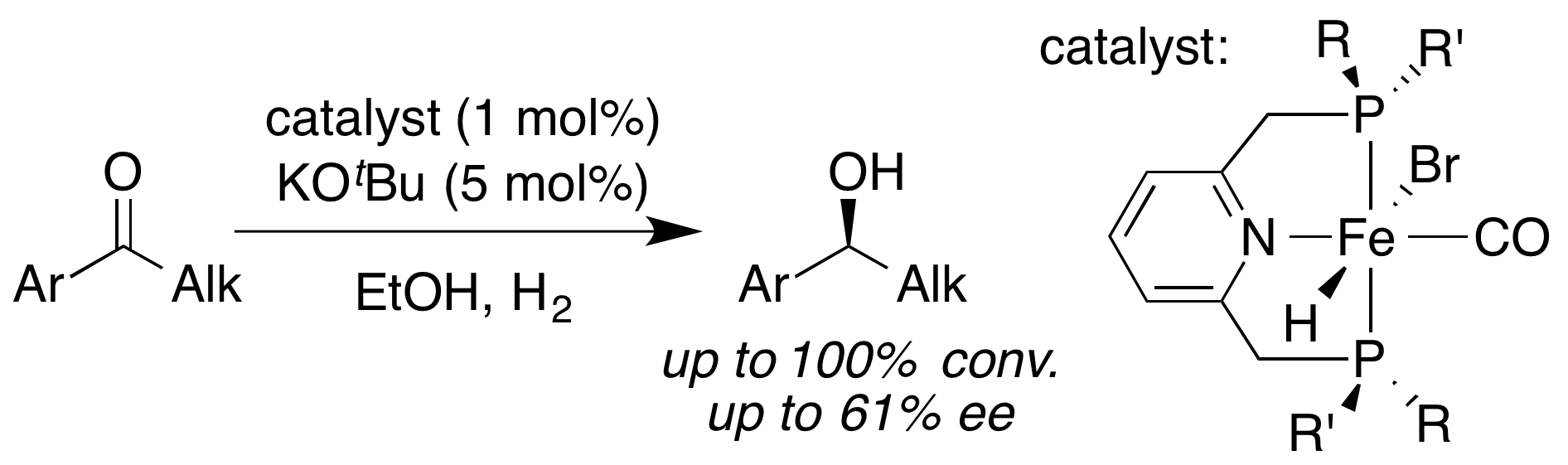
The corresponding bromocarbonylhydride complexes were prepared by addition of NaBHEt3 (1 equiv.) and either isolated or directly used in catalysis. They were active in the direct H2 hydrogenation of a variety of arylalkyl ketones under 50 bar of H2 pressure and in the presence of KOtBu as base. The enantioselectivity reached 61% ee with (S)-1-(2-naphthyl)ethanol. The origin of enantioselectivity was investigated using DFT calculations.
[1] A. Zirakzadeh, K. Kirchner, A. Roller, B. Stöger, M. Widhalm, R. H. Morris Organometallics 2016, 35, 3781.
[2] S. A. M. Smith, P. O. Lagaditis, A. Lüpke, A. J. Lough, R. H. Morris Chem. Eur. J. 2017, DOI: 10.1002/chem.201701254.