Helical Oligophenyl Geländer Molecules
Polycyclic aromatic compounds (PACs) have been of interest to fundamental researchers and material scientists since the concept of molecular chemistry was born. Particularly, chiral PACs have attracted much attention for material applications due to their unique physical properties (e.g. UV absorbance, semiconductance). In addition, their structural beauty brings an exciting and interesting challenge for chemists [1]. Recently, our group succeeded in synthesising a novel type of Geländer molecule with a terphenyl backbone and a bannister oligomer [2,3]. The success of this synthesis inspired us to further investigate this unique structure by extending our research into three new pathways. The first, is the synthesis of longer oligomers (Figure 1a), in order to achieve one full turnover of the helical structure and to determine, if the chiral information is further transferred to the next phenyl unit. Secondly, we designed a fully hydrogen-carbon Geländer oligomer (Figure 1b), to obtain a closer-packed system, we expect this to prohibit racemisation and allow the separation of enantiomers with a chiral packed HPLC column. Our ultimate goal and third pathway is to introduce a molecular platform. For instance a 9,9′-spirobifluorene group would provide the Geländer system with an anchoring group capable of immobilizing the helical Geländer molecule onto a metal surface (Figure 1c). This would lead to surface functionalization and investigations using scanning tunneling microscopy (STM).
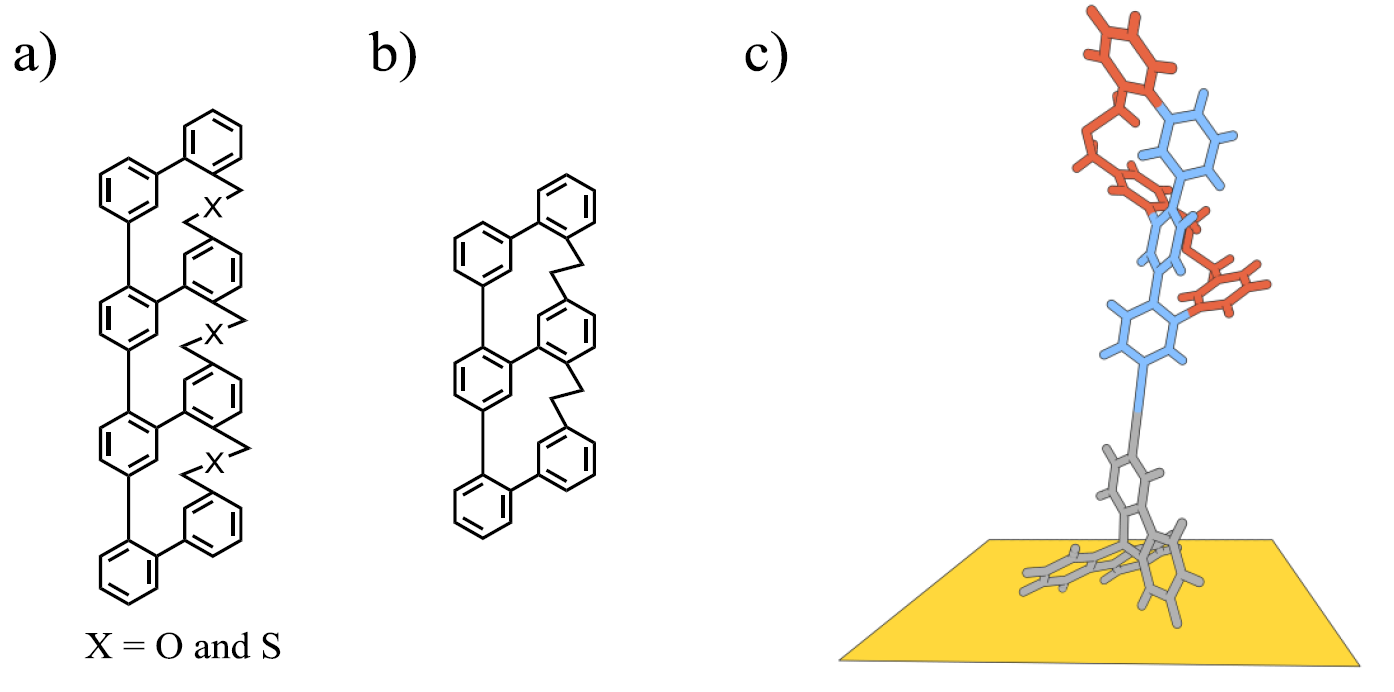
Figure 1: Three novel systems to investigate properties of chiral polycyclic aromatic compounds. a) Design of the higher-order oligomer species. b) Helical Geländer system containing only hydrogen and carbon atoms. c) 3D illustration of the immobilized Geländer molecule on a metal surface.
[1] Michel Rickhaus, Marcel Mayor, Michal Juríček, Chem. Soc. Rev., 2016, 45, 1542-1556.
[2] Michel Rickhaus, Linda Maria Bannwart, Markus Neuburger, Heiko Gsellinger, Kaspar Zimmermann, Daniel Häussinger, Marcel Mayor, Angew. Chem. Int. Ed., 2014, 53, 14587-14591.
[3] Michel Rickhaus, Oilver Unke, Rajesh Mannancherry, Linda Maria Bannwart, Markus Neuburger, Daniel Häussinger, Marcel Mayor, Chem. Eur. J., 2015, 21, 18156-18167.