Bimolecular Photoinduced Electron Transfer: the effect of the reorganization energy
Electron transfer (ET) is one of the simplest chemical reactions, as it does not require the formation or the breaking of a chemical bond. Moreover, photoinduced ET it is one of the most investigated photochemical reactions.1 ET processes are usually described theoretically in terms of the Marcus model, which connects the driving force of the reaction with its rate constant.2 The latter depends on several additional parameters such as the temperature, the reorganization energy and the electronic coupling between the initial and final states.
We will present a study focussing on the reorganization energy, which is composed of the contribution from the solvent, λs, and from intramolecular modes, λi. The effect of λs was investigated by measuring the dynamics of bimolecular photoinduced ET reactions in an apolar solvent and by comparison with previous results in a polar solvent.3 The effect of λi was studied by performing measurements with perdeuterated compounds.
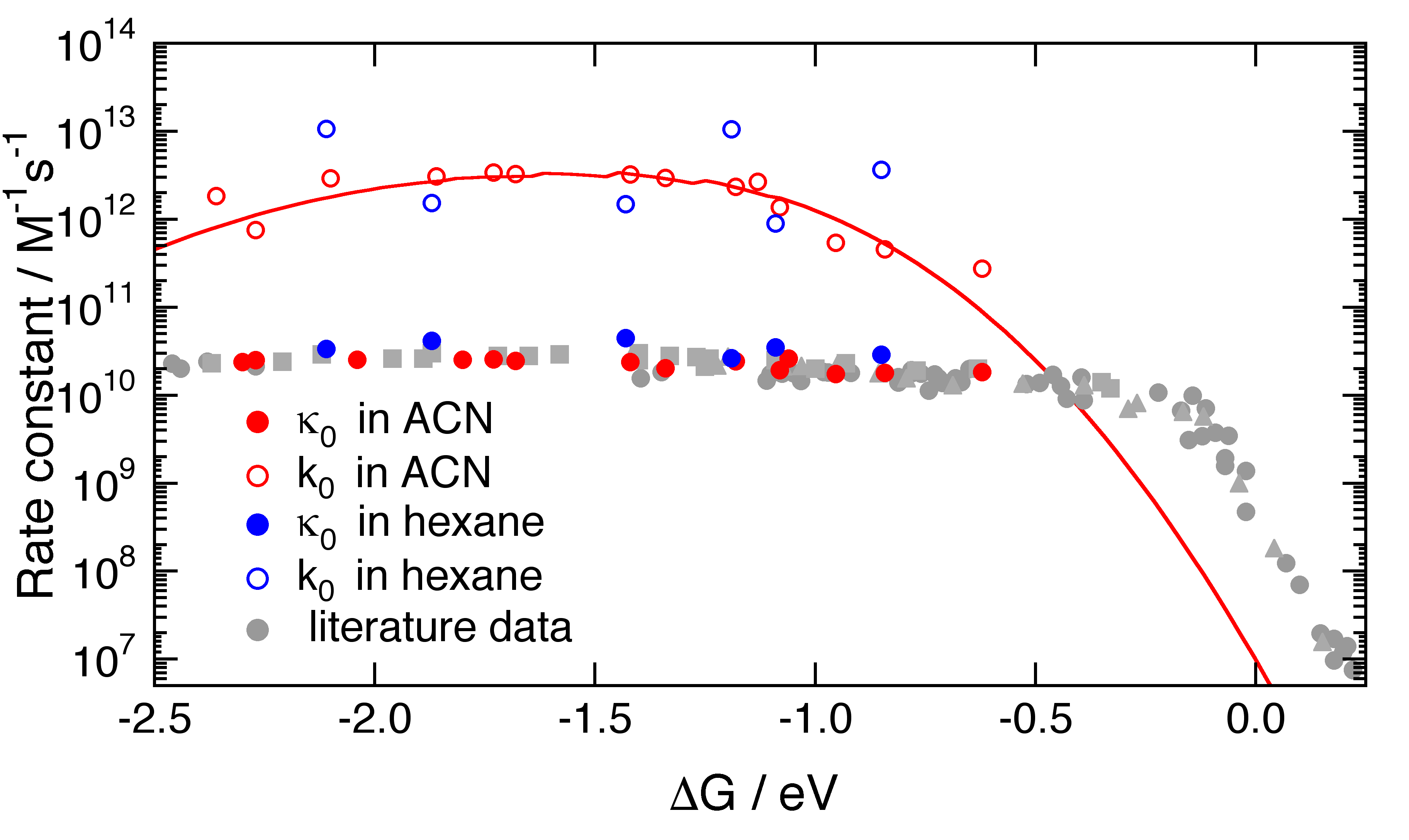
The above figure illustrates the driving force dependence of the ET rate constant in hexane and in acetonitrile (ACN). Here, k0 is the intrinsic, diffusion-free, bimolecular ET rate constant, whereas k0 is the diffusion-controlled rate constant. Surprisingly, the observed ET dynamics does not exhibit a significant dependence on the solvent polarity, despite very different λs. This result suggests that the intramolecular modes play a predominant role in the ET dynamics.
[1] Tatu Kumpulainen, Bernhard Lang, Arnulf Rosspeintner, Eric Vauthey, Chemical Reviews, 2017, DOI: 10.1021/acs.chemrev.6b00491
[2] R. A. Marcus, The Journal of Chemical Physics. 1956, 24, 966-78.
[3] Arnulf Rosspeintner, Gonzalo Angulo, Eric Vauthey, Journal of the American Chemical Society, 2014, 136, 2026-2032.