Structural Design, Computational Modeling, and Catalytic Pathways of Zn-Polyoxotungstate Catalysts
Polyoxometalates (POMs) are a fascinating class of metal-oxo clusters containing transition metal ions in their high oxidation states. Over the last 50 years, POMs have been widely explored for numerous applications in catalysis, as well as for their biomedical and magnetic features.[1] These metal-oxo clusters offer undeniable qualities for future applications, resulting from a remarkably wide structural and compositional diversity.
The number of POM architectures keeps growing steadily, and transition metal substituted POMs attract special attention as a rich reservoir for new catalyst developments. [2]
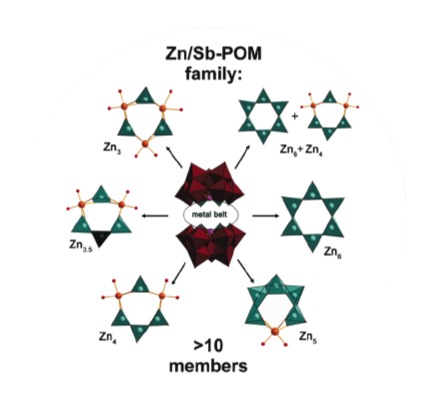
We introduce a new series of more than ten {ZnnNa6-n(B-a-SbW9O33)2} (n = 3-6) POMs with tunable Zn-cores that are sandwiched between two {B-α-SbW9O33} shells. This series shows high activity and selectivity in H2O2-assisted alcohol oxidation. Spectroscopic and kinetic studies suggest that the reaction proceeds via peroxotungstate intermediates, and strong experimental evidence for a radical mechanism is provided. These results are compared to a full scale solution-based computational modeling of the intermediates and transition states for [Zn6Cl6(SbW9O33)2]12- as a representative alcohol oxidation catalyst among the series.
[1] R. Sessoli, D. Gatteschi, A. Caneschi, M. A. Novak, Nature 1993, 365, 141.
[2] (a) Yang, Y.; Zhang, B.; Wang, Y. Z.; Yue, L.; Li, W.; Wu, L. X. J. Am. Chem. Soc. 2013, 135, 14500. (b) Suzuki, K.; Tang, F.; Kikukawa, Y.; Yamaguchi, K.; Mizuno. N. Angew. Chem. Int. Ed. 2014, 53, 5356.