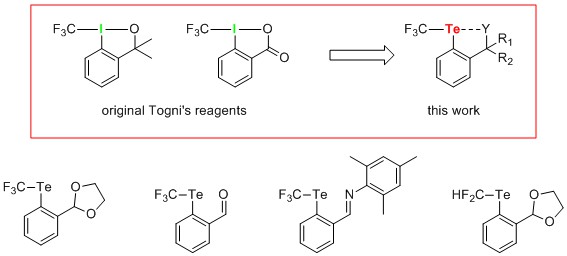
Synthesis of Hypervalent CF3 Tellurium Compounds
Hypervalent iodine (III) reagents are electrophilic trifluoromethylating agents commonly applied in organic syntheses. Since their introduction in 2006,[1,2] the parent structures have been subjected to numerous modifications. However to date, the central iodine atom has been retained in each instance.[3–5] Herein, we report for the first time a synthesis of unique compounds in which the iodine atom has formally been exchanged for tellurium. Thus, a series of CF3 tellurium (II) species differing in the functional group coordinated to the central atom has been obtained. All products have been fully characterized using HRMS and NMR spectroscopy, including advanced 1H125Te and 19F125Te correlation measurements. Furthermore, selected modifications performed after installing the CF3 group on the tellurium atom have been successfully carried out.[6]
[1] J. Charpentier, N. Früh, A. Togni, Chem. Rev. 2015, 115, 650–82.
[2] P. Eisenberger, S. Gischig, A. Togni, Chem. Eur. J. 2006, 12, 2579–86.
[3] K. Niedermann, J. M. Welch, R. Koller, J. Cvengroš, N. Santschi, P. Battaglia, A. Togni, Tetrahedron 2010, 66, 5753–5761.
[4] N. Santschi, R. C. Sarott, E. Otth, R. Kissner, A. Togni, Beilstein J. Org. Chem. 2014, 10, 1–6.
[5] V. Matoušek, J. Václavík, P. Hájek, J. Charpentier, Z. E. Blastik, E. Pietrasiak, A. Budinská, A. Togni, P. Beier, Chem. Eur. J. 2016, 22, 417–24.
[6] E. Pietrasiak, A. Togni, manuscript in preparation