Radical geometry: a key to efficient Dynamic Nuclear Polarization.
Dynamic nuclear polarization (DNP) is becoming an essential tool to dramatically improve the sensitivity of numerous experiments in nuclear magnetic resonance (NMR) including those involving the elucidation of surface structure of advanced materials1 and the characterisation of polymorphs in organic solids2. The method is based on the transfer of polarization from electron to nuclear spins by saturation of the electron spin transitions. The upper limit of transferable polarization, at equilibrium, is dictated by the ratio of the gyromagnetic values of electron and nuclear spins: about 660 for 1H and even more for other low abundant nuclei such as 13C, 15N. The primary parameter to perform an efficient DNP experiment remains by far the choice of the radical3. Several factors have been identified to affect the efficiency of the polarising agent: rigidity, bulkiness, relaxation properties to name a few.
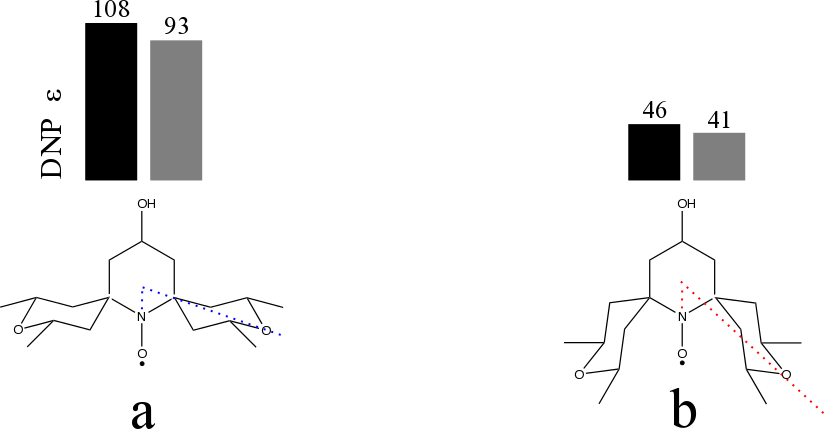
Fig. 1: Nitroxide monoradicals used as polarizing agents for MAS ssDNP experiments at 9.4T, ~100 K, MAS=8000 Hz. The displacement of the cyclohexane sidearms from the nitroxide axis is larger for the open than for the closed configuration (panel a) and b) respectively). The rectangles indicate the MAS ssDNP enhancement obtained via 1H-13C cross polarization when the solvent used is glycerol-d8:D2O:H2O (black) and TCE (grey).
Here we show for a set of nitroxide mono- and bi-radicals how the local geometric conformation, especially in the vicinity of the unpaired electron spin, leads to considerably different enhancement factors for MAS ssDNP (Magic Angle Spinning solid state DNP) experiments conducted at 9.4 T and ~100K in 1,1,2,2-tetrachloroethane (TCE), glycerol-d8:D2O:H2O, and DMSO-d6:D2O:H2O. Given the impact on the experimental enhancement factor we include the local geometry among the set of important variables to consider in the design of new polarising species. The open configuration (Fig 1a) leads always to a better DNP enhancement either for the nitroxides mono- and bi-radicals irrespective of the solvent used. The different DNP performance between open and closed is currently accounted for by postulating a difference in solvent accessibility to the unpaired electron caused by the bulky substituents.
[1] A. J. Rossini, A. Zagdoun, M. Lelli, A. Lesage, and C. Cop, 2013,
[2]A. C. Pinon, A. J. Rossini, C. M. Widdifield, D. Gajan, and L. Emsley, Mol. Pharm., 2015, 12, 4146–4153.
[3] D. J. Kubicki, A. J. Rossini, A. Purea, A. Zagdoun, O. Ouari, P. Tordo, F. Engelke, A. Lesage, and L. Emsley, J. Am. Chem. Soc., 2014, 136, 15711–15718.