A Tethering Strategy for the Synthesis of Vicinal Aminoalcohols
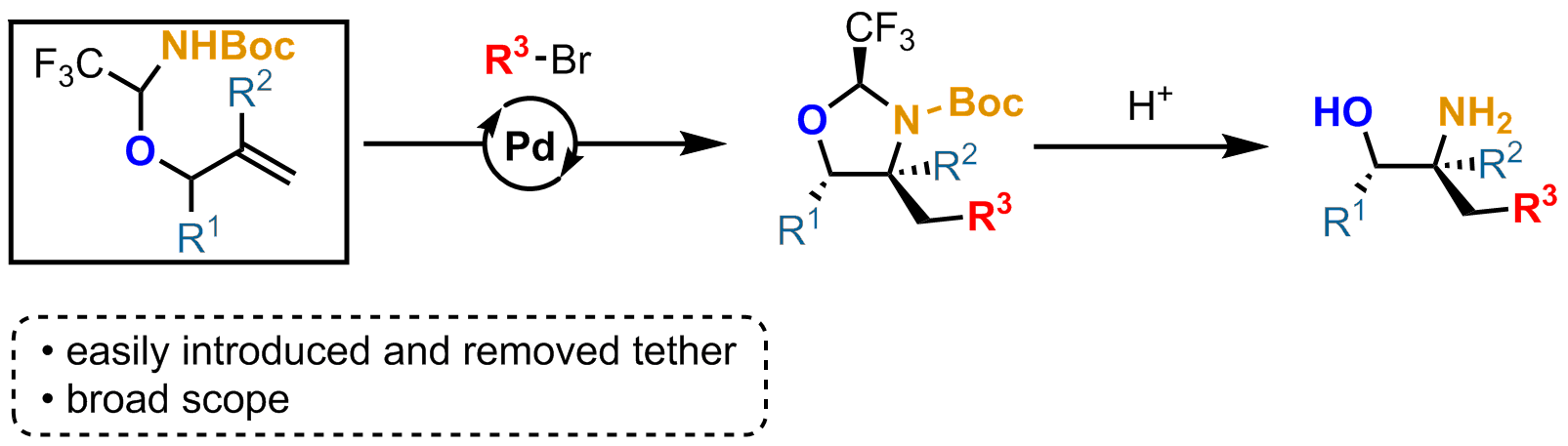
Vicinal amino-alcohols are a common structural motif that can be found in a wide array of biologically active natural compounds, ligands, catalysts and chiral auxiliaries.[1] Therefore, efficient methods for their synthesis are necessary. The use of removable tethers combined with Pd-catalyzed olefin functionalization has known intense developments in recent years and has proved to be an efficient strategy for the installation of new functionalities on an alkene in a 1,2 relationship.[2] Recently, our group has introduced (hemi)aminal tethers derived from trifluoroacetaldehyde for the carboetherification and carboamination of allylic amines to give aminoalcohols and diamines.[3] Herein, we would like to report the successful implementation of this strategy to allylic alcohols. The synthesis of a stable hemiaminal could be achieved, followed by a Pd-catalyzed carboamination that could install concomitantly a C-N bond and a highly valuable C-C bond.[4] A wide variety of groups could be introduced to form functionalized oxazolidines that could then deliver the free amino alcohols under acidic hydrolysis.
[1] Stephen C. Bergmeier, Tetrahedron, 2000, 56, 2561-257.
[2] a) Zachary J. Garlets, Derick R. White, John P. Wolfe, Asian J. Org. Chem., 2017, doi:10.1002/ajoc.201600577; b) Ugo Orcel, Jerome Waser, Chem. Sci., 2017, 8, 32-39.
[3] a) Ugo Orcel, Jerome Waser, Angew. Chem., Int. Ed., 2015, 54, 5250-5254; b) Ugo Orcel, Jerome Waser, Angew. Chem., Int. Ed., 2016, 55, 12881-12885.
[4] Bastian Muriel, Ugo Orcel, Jerome Waser, Manuscript in preparation.