Gram–Scale Biomimetic Synthesis of Psiguadial B
In recent years a growing number of synthetic chemists have begun to embrace the philosophies of efficient synthesis (e.g. atom, step, redox economy, etc.), indicative of a shift in focus away from purely the feasibility of a synthetic route, but rather towards considering the overall efficiency of a strategy.[1] Generally speaking, reactions that form multiple chemical bonds lead to a more rapid generation of structural complexity versus traditional stepwise approaches, ultimately resulting in improved syntheses.
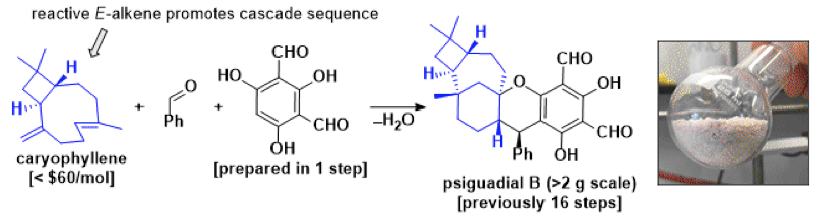
Herein we present the first preparative synthesis of the complex meroterpenoid psiguadial B, a potent inhibitor of human hepatoma cell growth.[2] Psiguadial B was previously prepared in 16 steps, and in only milligram quantities, thus limiting further biological appraisal of the natural product.[3] Combined computational and experimental investigations from our research group suggest the biosynthesis of the natural product proceeds through a Michael addition between caryophyllene and a reactive ortho-quinone methide, followed by two sequential intramolecular cationic cyclization events. Through these considerations we have been able to develop a biomimetic synthesis of the psiguadial B via a three component coupling of caryophyllene, benzaldehyde and diformylphloroglucinol, enabling access to the molecule on gram-scale, and in only 2 steps.
[1] Tanja Gaich, Phil S. Baran, J. Org. Chem. 2010, 75, 4657–4673.
[2] Meng Shao, Ying Wang, Zhong Liu, Dong-Mei Zhang, Hui-Hui Cao, Ren-Wang Jiang, Chun-Lin Fan, Xiao-Qi Zhang, He-Ru Chen, Xin-Sheng Yao, and Wen-Cai Ye, Org. Lett. 2010, 12, 5040−5043.
[3] Lauren M. Chapman, Jordan C. Beck, Linglin Wu, aSarah E. Reisman, J. Am. Chem. Soc. 2016, 138, 9803−9806.