On the Generation and the Properties of the F3C• Radical
"Introduction of the trifluoromethyl group into organic molecules can lead to drastic and desirable changes in chemical and physical properties" - Statements along this line regularly serve as motivation for application-driven research. However, within the framework of this approach, primarily the functionalized product is of interest and relatively little attention is being paid to the enabling underlying mechanistic scheme. For example, while the addition of the electrophilic (?) F3C• radical to arenes is undoubtedly an important process, it is most often depicted as an irreversible step - this constitutes a severe misconception.
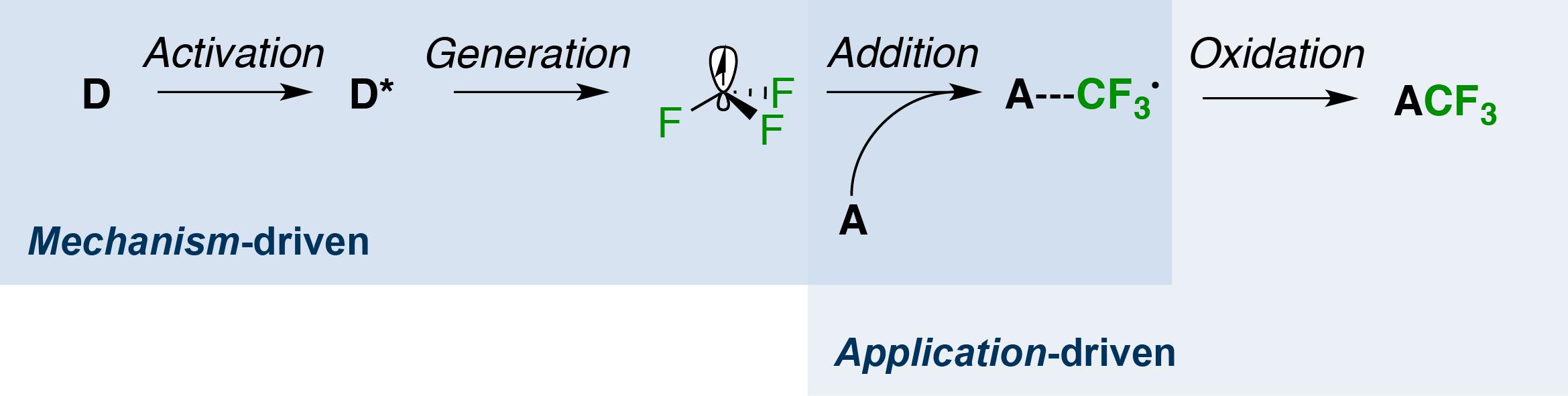
Subject of the present contribution are the various facets of our mechanism-driven approach towards studying the generation of the trifluoromethyl radical and elucidating its properties. In this respect, it is important to realize that this reactive intermediate has to be accessed from a stable precursor D by a precursor-matched set of activating conditions, which then lead to an intermediate D*. Subsequently, the latter will decay and release the species of interest. After addition of the trifluoromethyl radical to an acceptor A, a final single-electron oxidation will provide the stable product. In the context of this genreal mechanistic scheme, we will discuss:
(1) The limitations of oxidative and reductive activation strategies of commercially available F3C• precursors (trifluoroacetate, Langlois reagent and Togni's reagent) with primary radiolysis products (H•, HO•, e-).[1]
(2) The additive-free, thermal activation of the hypervalent iodine-based Togni reagent as investigated by gas phase thermolysis in standard GC-MS equipment and the extension to a laboratory scale setup.[2]
(3) The degree of reversibility of F3C• addition to an aromatic model system and how this approach can be generalized to arrive at an experimental electrophilicity scale for radicals.[3]
[1] a) Nico Santschi*, Thomas Nauser, J. Fluorine Chem. 2017, accepted. b) Nico Santschi*, Thomas Nauser, unpublished results.
[2] a) Nico Santschi*, Benson J. Jelier, Thomas Nauser, submitted. b) Nico Santschi*, Benson J. Jelier, submitted. [3] Nico Santschi*, Thomas Nauser, submitted.