Azetidin-1-yl substituents to tune the photochemistry of photoactive molecules
The ability to tune the photochemical evolution of a chromophore through structural modification is crucial to design photoresponsive materials for biological or chemical applications. There have been reports of remarkable increases in the fluorescence quantum yields of several dyes upon swapping a dialkylamino substituent for an azetidin-1-yl group (Figure 1a),[1,2] possibly arising from the inhibition of the formation of Twisted Intramolecular Charge Transfer (TICT) states (Figure 1b).[1,2] A photoprotecting group (PPG) is a chemical moiety that masks the activity of the compounds it is bound to, and that can be removed upon excitation with one or two photons.[3] 7-Dialkylamino-4‑methyl-coumarin derivatives have been widely used as PPGs, even though the photorelease efficiencies (φPA) of the caged compounds tend to be moderate to low (usually below 20%).[3,4] We reasoned that the formation of a TICT state could be a competitive process for the photorelease reaction of 7-dialkylamino coumarins, and that substitution with an azetidin-1-yl group might prove beneficial. To test this hypothesis, we prepared a series of caged esters of 7-diethylamino (1a-b) and 7-azetidin-1-yl (2a-b) 4-methyl coumarin derivatives (Figure 1c) and measured their φPA (Figure 1d). The azetidin-1-yl substituted coumarins showed a higher φPA than the 7-diethylamino derivatives, with values comparable to julolidine‑fused derivatives (3a-b), that cannot form a TICT state (Figure 1d). These results constitute a proof-of-principle demonstration of the feasibility to exploit the inhibition of TICT states formation by azetidines to enhance the quantum yield of photochemical processes that are in kinetic competition with TICT states formation. We are actively investigating other applications of this substitution, to broaden the scope even further.
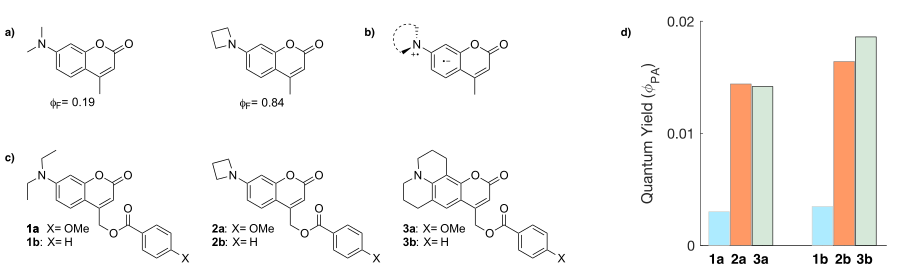
Figure 1. (a) Reported increase in fluorescence quantum yield (φF) for 7-(azetidin-1-yl)-4-methyl coumarin. (b) TICT state for a generic 7-dialkylamino-4-methyl coumarin. (c) Structures of the 4-methyl-coumarin benzoates studied. (d) Measured quantum yield of photorelease (φPA).
[1] J. B. Grimm, B. P. English, J. Chen, J. P. Slaughter, Z. Zhang, A. Revyakin, R. Patel, J. J. Macklin, D. Normanno, R. H. Singer, T. Lionnet, L. D. Lavis, Nat. Methods 2015, 12, 244–253.
[2] X. Liu, Q. Qiao, W. Tian, W. Liu, J. Chen, M. J. Lang, Z. Xu, J. Am. Chem. Soc. 2016, 138, 6960–6963.
[3] P. Klán, T. Šolomek, C. G. Bochet, A. Blanc, R. Givens, M. Rubina, V. Popik, A. Kostikov, J. Wirz, Chem. Rev. 2013, 113, 119–191.
[4] R. S. Givens, M. Rubina, J. Wirz, Photochem. Photobiol. Sci. 2012, 11, 472–488.