Using puled EPR dipolar spectroscopy for the coarse-grained localisation of a residue in the intrinsically disordered domain of hnRNP A1
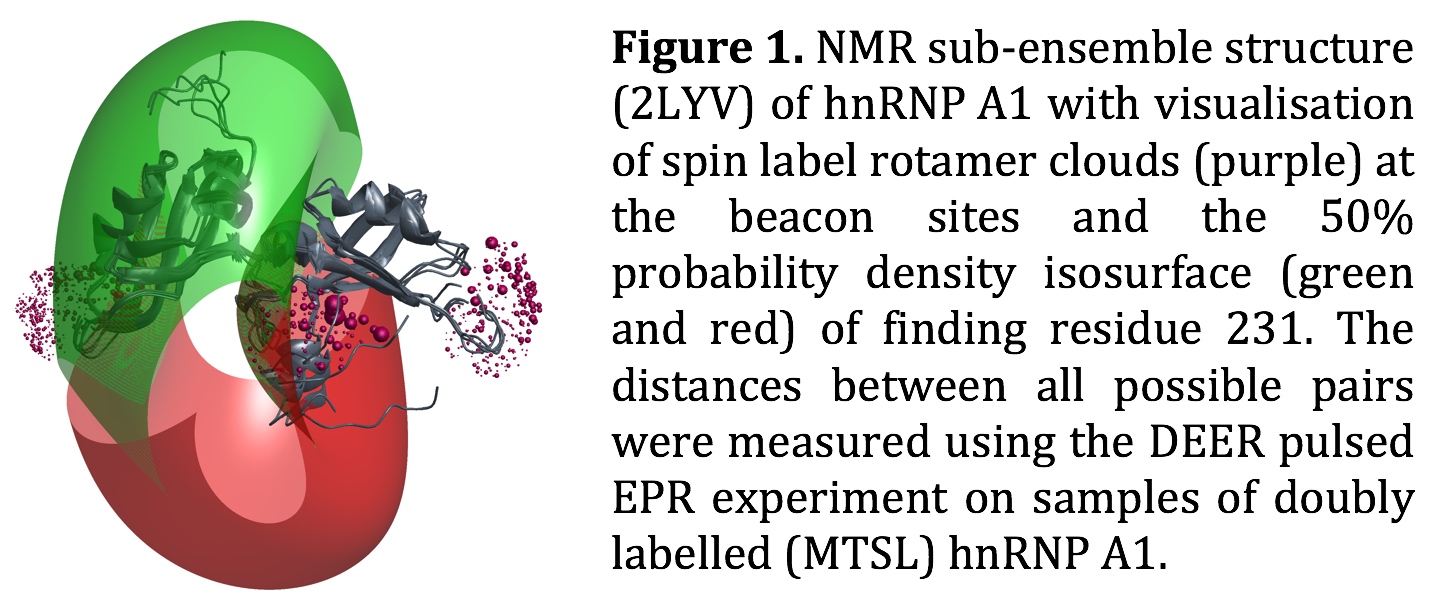
The regulatory function of the human protein hnRNP A1 in splicing is believed to be mediated by multimerisation of individual hnRNP A1 units on long RNAs via an intrinsically disordered C-terminal domain [1]. The two N-terminal domains of hnRNP A1 are known to fold in a well-defined structure, which has been solved by X-ray crystallography, and more recently by solution NMR (pdb: 2LYV). The high degree of flexibility of the C-terminal domain, however, makes it inaccessible for both structural techniques. Using site-directed spin labelling, we determine distance distributions from residues in the structured domains, termed beacon sites, to individual residues in the disordered domain using the DEER pulsed EPR technique [2]. Together with a set of distances measured between the beacon sites themselves, it is then possible to calculate a spa-tial distribution of the position of a given residue in the disordered domain, which can be visualised as an isosurface of the probability density [3]. This analysis approach is included in the current version of the modelling software MMM [4].
Support: This work is supported by SNF grant 200020 157034.
[1] H. L. Okunola, A. R. Krainer, Cooperative-Binding and Splicing-Repressive Properties of hnRNP A1, Molecular and Cellular Biology, 2009, 29, 5620–5631
[2] G. Jeschke, DEER Distance Measurements on Proteins, Annual Review of Physical Chemistry, 2012, 63, 419–446
[3] G. Jeschke, Ensemble models of proteins and protein domains based on distance distribution restraints, Proteins, 2016, 84, 544–560
[4] G. Jeschke, Y. Polyhach, E. Bordignon, Multiscale Modeling of Macromolecules—MMM, 2010, ETH Zurich (http://www.epr.ethz.ch/software.html)