Radical Reaction of Sulfonyl Chloride in Access to Vinyl Sulfones
Vinyl sulfones are extensively used as a building blocks for total synthesis of natural products[1]. They can be prepared by olefination reactions[2], elimination from α- or β-substituted sulfones[3], oxidation of vinyl sulfides[4] or others.
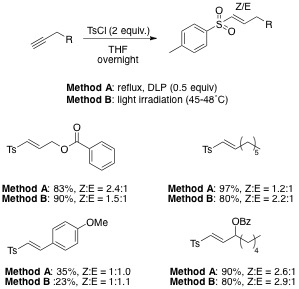
Their preparation from terminal alkynes is highly attractive. However, such transformations have only been reported via hydrometallation process[5].
Herein we describe selective introduction of vinyl sulfones. We generate the sulfonyl radical from p-toluenesulfonyl chloride and add it onto alkyne.
[1] Riccardo Piccardi, Philippe Renaud, P. Eur. J. Org. Chem. 2007, 4752-4757
[2] Vijay Nair, Anu Augustine, T. D. Suja, Synthesis 2002, 2259-2265
[3] Hao Qian, Xian Huang, Synlett 2001, 1913-1916
[4] Xian Huang, De-Hui Duan, Weixin Zheng, J. Org. Chem. 2003, 68, 1958-1963
[5] De-Hui Duan, Xian Huang, Synlett, 1999, 3, 317–318