Fluorophore guided RNA modeling
Non-coding RNAs engage in numerous cellular processes at various stages of gene expression. As a spectroscopic ruler [1] single-molecule FRET is a versatile tool to investigate the folding of RNA [2]. Yet, interconversion of transfer efficiencies and absolute distances between a donor and an acceptor fluorophore requires thorough control over both the dyes' photophysics as well as their orientation within the macromolecule [3]. We make use of the photoisomerization propensity of carbocyanines [4] to sense perturbations in their local environment. Changes to the torsional flexibility of the polymethine scaffold are probed by time-resolved fluorescence anisotropy and RNA-induced fluorescence enhancement (RIFE) [5]. The constrained dye mobility due to entrapment on the RNA surface is evaluated by means of all-atom molecular dynamics simulations using the exon and intron binding site (EBS1/IBS1) of a group II intron [6] as a model of an RNA tertiary contact [5]. These atomistic simulations give access to the dye-linker dynamics which are key to defining the volume accessible to the fluorophore in the context of the RNA.
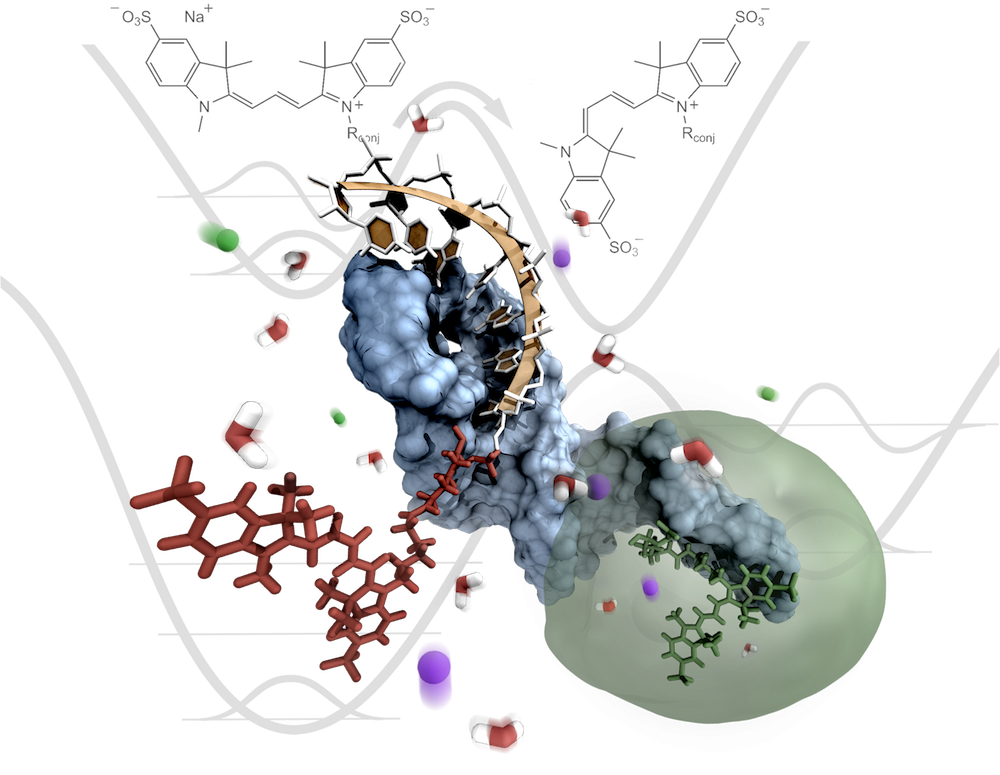
Building on a refined description of the dyes, a set of de novo modeled RNA structures is then filtered against experimentally derived distance distributions of one or multiple FRET pairs. Imposing such restraints on different parts of the RNA confines the search space while at the same time pinpointing regions of increased flexibility. We validate this integrative modeling approach by reconstituting the solution structure of the EBS1 hairpin motif in silico. After all, the combination of smFRET and molecular modeling will open up new avenues to explore the world of long non-coding RNAs whose three-dimensional fold is yet to be discovered.
[1] Lubert Stryer, Richard. P. Haugland, Proc. Natl. Acad. Sci. USA, 1967, 58, 719-726.
[2] Richard Börner, Danny Kowerko, Helena Guiset-Miserachs, Michelle F. Schaffer, Roland K.O. Sigel, Coord. Chem. Rev., 2016, 327-328, 123-142.
[3] Benjamin Schuler, Everett A. Lipman, Peter J. Steinbach, Michael Kumke, William A. Eaton, Proc. Natl. Acad. Sci., 2005, 102, 2754-2759.
[4] Marcia Levitus, Suman Ranjit, Q. Rev. Biophys, 2011, 44, 123-151.
[5] Fabio D. Steffen, Roland K.O. Sigel, Richard Börner, Phys. Chem. Chem. Phys., 2016, 18, 29045-29055.
[6] Danny Kowerko, Sebastian L.B. König, Miriam Skilandat, Daniela Kruschel, Mélodie C.A.S. Hadzic, Lucia Cardo, Roland K.O. Sigel, Proc. Natl. Acad. Sci., 2015, 112, 3403-3408.