Dopant Screening of Ceria-Based Materials for Solar Thermochemical Two-Step CO2-Splitting
We consider the solar-driven 2-step thermochemical cycle for splitting H2O and CO2 via CeO2-based redox reactions[1]. In the first endothermic step, ceria is thermally reduced using concentrated solar energy by releasing O2 (Eq. 1). In the second exothermic step (Eq. 2a and 2b), reduced ceria reacts with H2O and/or CO2 to generate H2 and/or CO, respectively. The fuel yield is proportional to the oxygen exchange capacity (OEC), which can be improved with cationic doping.
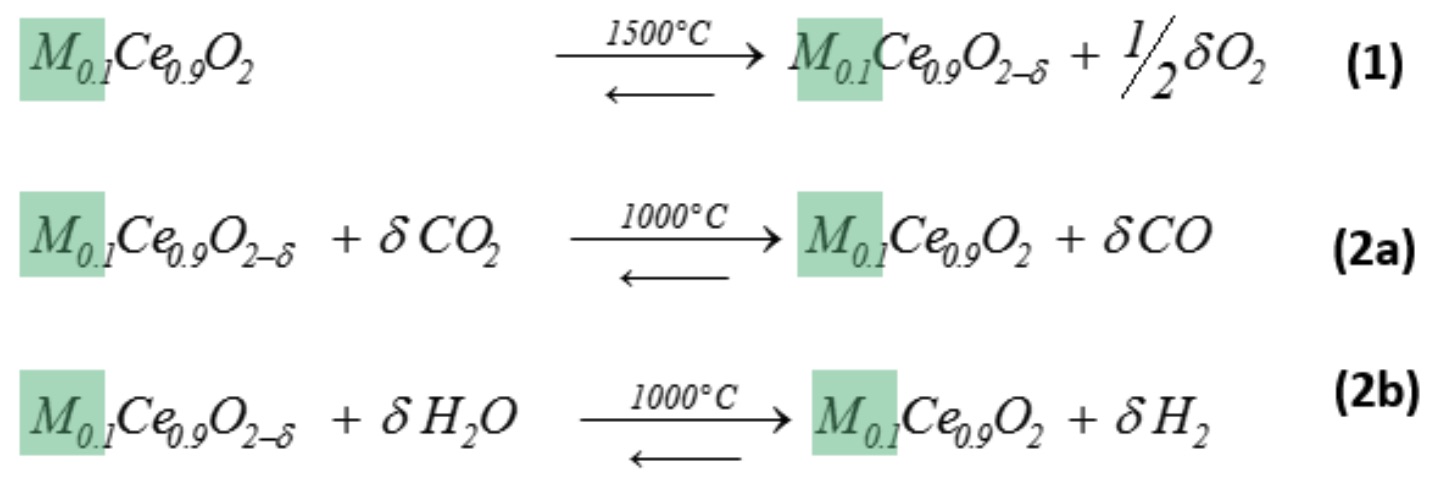
Starting from our earlier work,[2] we investigate all possible tetravalent dopants of the periodic table because of their potential to yield higher OEC values due to flexible vacancy formation processes. The doped ceria samples were analyzed with a wide range of analytical techniques (e.g. PXRD, TG, ICP-MS, SEM-EDX, XAS). We found a correlation between the effective ionic radius and the OEC that was supported by DFT calculations. The tetravalent dopants with best redox performance were tested for their high temperature long-term stability in thermogravimetric cycling experiments.
[1] W.C. Chueh, C. Falter, M. Abbott, D. Scipio, P. Furler, S.M. Haile, A. Steinfeld, Science 2010, 330, 1797.
[2] J.R. Scheffe, R. Jacot, G.R. Patzke, A. Steinfeld, J. Phys. Chem. C 2013, 117, 24104.