General Access to 1,1 and 1,2 Azidolactones from Alkenes using Hypervalent Iodine Reagents
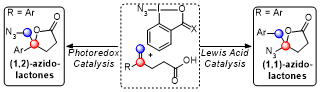
Amino lactones[1] and amino alcohols[2] represent important classes of compounds, which have crucial applications in natural products synthesis and in medicinal chemistry. In recent years, neutral cyclic hypervalent iodine reagents benziodoXoles have emerged as privileged tools in group-transfer chemistry. In addition, being stable crystalline compounds, they are easy to handle and user friendly.[3] In particular, AzidoBenziodoXolones (ABX) and AzidoDimethylBenziodoXoles (ADBX) have demonstrated high efficiency in the transfer of azido group onto organic molecules using metal catalysis. [3] Herein, we describe a versatile synthesis of azidolactones through the cyclization of carboxylic acids onto alkenes, based on photoredox and palladium catalysis. (1,1) and (1,2) azido lactones can be selectively synthesized. Modulating the properties of the catalyst and the benziodoxol(on)e reagent serving as azide source led to either a radical or Lewis Acid mediated process and favored formation of only one of the possible regioisomers starting from the same starting material. These transformations have been carried out under mild conditions using a low catalyst loading and gave access to a large scope of azido lactones.[4]
[1] a) G. Blaskó, D. J. Gula, M. Shamma, J. Nat. Prod. 1982, 45, 105; b) M. Bös, H. Stadler, J. Wichmann, F. Jenck, J. R. Martin, J.-L. Moreau, A. J. Sleight, Helv. Chim. Acta 1998, 81, 525; c) C. Kaiser, C. J. Spagnuolo, T. C. Adams, V. H. Audia, A. C. Dupont, H. Hatoum, V. C. Lowe, J. C. Prosser, B. L. Sturm, L. Noronha-Blob, J. Med. Chem. 1992, 35, 4415; d) Y. Shimojima, H. Hayashi, J. Med. Chem. 1983, 26, 1370.
[2] a) S. C. Bergmeier, Tetrahedron 2000, 56, 2561; b) T. J. Donohoe, C. K. A. Callens, A. Flores, A. R. Lacy, A. H. Rathi, Chem. Eur. J. 2011, 17, 58.
[3] Y. Li, D. P. Hari, M. V. Vita, J. Waser, Angew. Chem. Int. Ed. 2016, 55, 4436.
[4] S. Alazet, F. Le Vaillant, S. Nicolai, Th. Courant, J. Waser, 2017, submitted for publication.