Modeling Heterogeneous Catalysts by First Principles
Metallic nanoparticles supported on oxides are one of the most important industrial heterogeneous catalysts. They are used in the ammonia synthesis, the formation of hydrocarbons from CO/H2 (Fischer-Tropsch Synthesis), the reforming of methane or the water-gas shift reaction. In most cases, the evaluation of the reactivity of these systems via ab initio simulations is performed on a metallic facet (either a terrace of a step), usually neglecting the effect of the oxide support. While this approach allows the screening of different catalysts for a particular reaction at reasonable computational cost, it is not always able to explain how such systems work at atomic level. In this contribution, we will show how the use of realistic models for oxide supported nanoparticles can be key to properly describe the active site(s) of such systems and to rationalize experimental observations. In particular, we will illustrate how this approach combined with bonding analysis provides an additional explanation of the particle size effect in the Fischer-Tropsch reaction catalyzed by Ru nanoparticles; i. e. why bigger particles are more reactive towards CO than smaller ones.[1] Moreover, we will show how the inclusion of the support provides new interface sites, which are active in the methanol synthesis (from CO2 and H2) and the water gas-shift reaction catalyzed by oxide-supported Cu and Ni nanoparticles, respectively.[2,3]
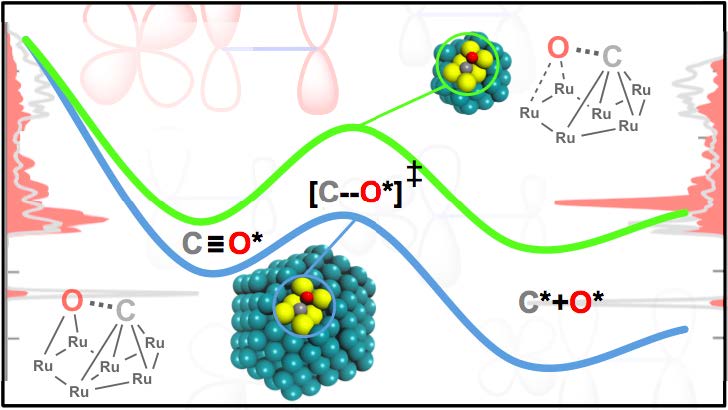
[1] Lucas Foppa, Christophe Copéret, Aleix Comas-Vives, J. Am. Chem. Soc., 2016, 138, 16655-16668.
[2] Kim Larmier, Wei-Chih Liao, Shohei Tada, Erwin Lam, René Verel, Atul Bansode, Atsushi Urakawa, Aleix Comas-Vives, Christophe Copéret. Angew. Chem. Int. Ed., 2017, 129, 2358-2363.
[3] Marius-Christian Silaghi, Aleix Comas-Vives, Christophe Copéret ACS Catal., 2016, 6, 4501-4505.