The Influence of Phosphonic Acid Protonation State on the Efficiency of Bis(diimine)copper(I)-based Dye Sensitized Solar Cells
The photoactive component of a Dye-Sensitized Solar Cell (DSC) consists of a dye molecule bound (either electrostatically or covalently) to a mesoporous TiO2 surface through functional groups such as phosphonic acids.[1] Here, we investigate how the protonation state of such phosphonic acids affects their ability to anchor to TiO2 substrates, and how the operational parameters of the resultant DSCs are influenced in turn.
Stepwise titration of bases into a solution of LH4 alters the ligands protonation state and the nature of its associated cations to give [LHn][X]n-4 (Fig. 1). The formation of Cu(I) dye molecules is achieved by sequentially exposing TiO2 electrodes to [LHn][X]n-4, followed by the homoleptic Cu(I) complex of the ancillary ligand. Our results demonstrate that the addition of a small amount of base to LH4 (~1 eq) can afford up to a 30% increase in DSC efficiency. These results are rationalized through consideration of proton / cation transfer from the ligand to the surface, deprotonation of surface hydroxides, and changes in the photochemical and electrochemical properties of ligand LH4 upon deprotonation.
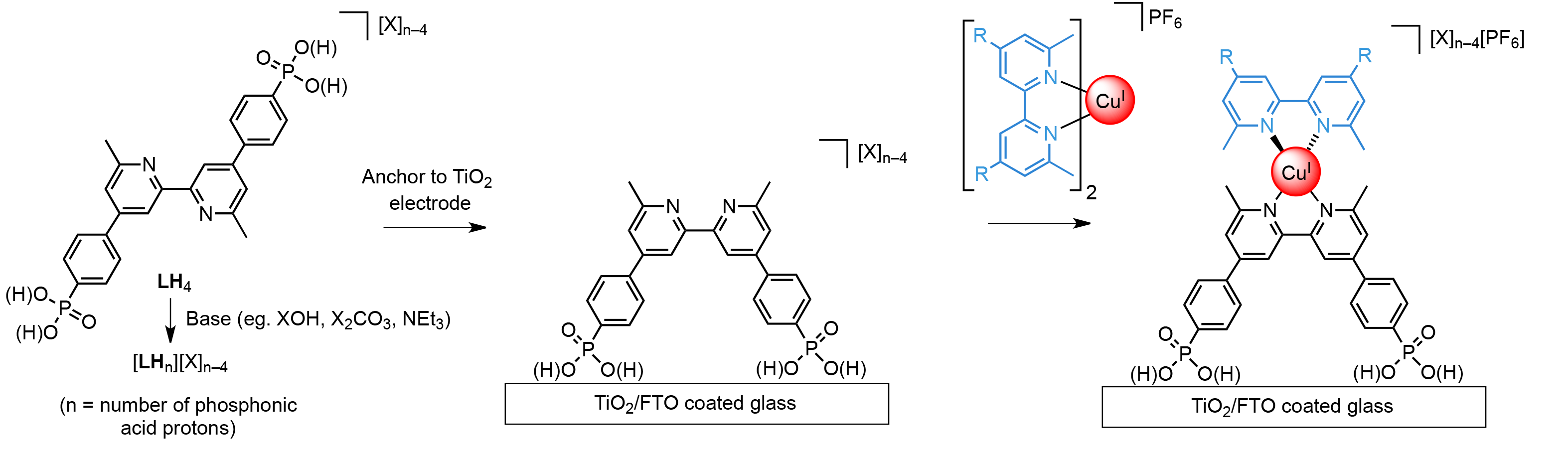
Figure 1. The sequential formation of a bis(diimine)copper(I) dye on a TiO2 electrode using phosphonic acids in different protonation states.
[1] C. E. Housecroft, E. C. Constable, Chem. Soc. Rev., 2015, 44, 8386–8398.