Controlled deposition of metal oxide layers by non-hydrolytic sol-gel for tailored acid sites generation and beyond
Bulk materials are omnipresent in catalysis due to their ease of production, but they often suffer from low selectivity and stability due to the lack of precise design of their surface functionalities. We present a new technique for the controlled deposition of metal oxides layers, where precise tuning of material surface functionalities yields optimal performances for various applications.
The method is based on non-hydrolytic sol-gel chemistry using mixtures of metal alkoxides and metal halides. For instance, addition of a 1/3 mol/mol mixture of Ti(iPrO)4 and TiCl4 to a toluene suspension of SiO2 spheres led to the formation of a thin conformal TiO2 overcoat with tunable porosity (Fig. 1, left). Thickness can be adjusted by the number of deposited layers with a growth rate of 0.5 nm/layer. Furthermore, this method allows deposition of TiO2 layers inside the pores of mesoporous silica SBA-15 while maintaining 2D hexagonal structure and high porosity (Fig. 1, right). The mixed oxide created at the interface generates medium strength Lewis acid sites which catalyze 1-phenylethanol dehydration to styrene. High Lewis acid sites density with medium acid strength was obtained with 6 TiO2 layers which lead to the highest yields of styrene and the highest catalyst stability. In contrast, a zeolitic catalyst often used for this reaction (HZSM-5) deactivates due to polystyrene formation on strong Lewis acid sites. The technique has also been employed to overcoat supported metal nanoparticle catalysts leading to metal-acid bifunctional catalysts that are highly selective for 3-propylguaiacol hydrodeoxygenation, a key transformation for lignocellulosic biomass valorization. Finally, an analogous method was developed for deposition of alumina, yielding strong acid sites after deposition on silica substrates. We anticipate that the method reported can be used for the rational design of catalytic materials used in a wide range of applications.
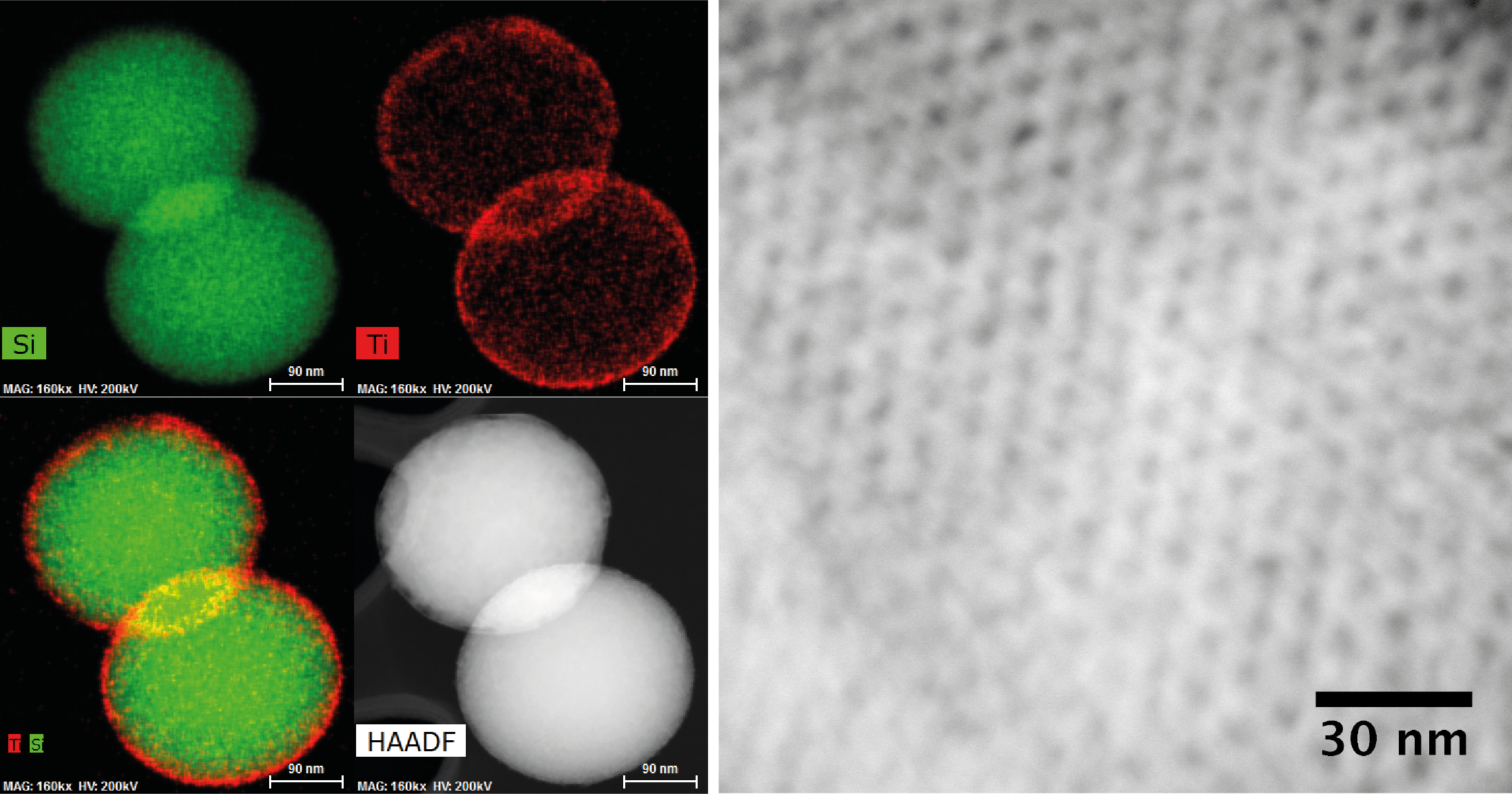
Fig. 1. Left: EDS mapping and HAADF STEM image of SiO2 sphere after deposition of 10 layers TiO2 (Si: green; Ti: red). Right: HAADF STEM image of SBA-15 after deposition of 10 layers TiO2.