Mechanically triggered fluorescent changes in polymers
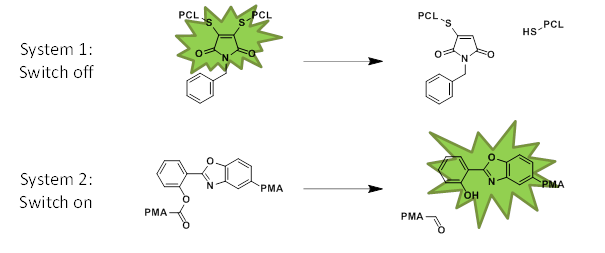
Despite an increasing number of studies that have investigated mechanochemical effects in polymers, the number of polymers whose fluorescence characteristics change upon exposure to mechanical stress is still limited. Here, we report on two families of mechanoresponsive polymers whose fluorescence can be mechanically turned on or off, respectively. The fluorescent dithiomaleimide (DTM) motif was used as a mechanically responsive moiety whose fluorescence can be turned off upon mechanically induced chain cleavage. This mechanophore was incorporated into poly(ε-caprolactone)s (PCL) of different molecular weights through ring opening polymerization initiated from a DTM dihydroxy derivative. This strategy allows us to position the DTM in the middle of a PCL chain and further study its response to ultrasound-induced mechanical stress. Indeed, when solutions of high molecular-weight PCL-DTM were exposed to ultrasound, the fluorescence intensity and molecular weight decreased according to first-order kinetics with comparable rate constants. A substantial decrease of the fluorescence intensity was also observed when solid samples of the high molecular-weight PCL-DTM were grinded. The data thus demonstrate the usefulness of the DTM motif as mechanophore that displays fluorescence turn-off upon mechanochemically induced scission. On the other hand, a hydroxy-benzoxazole derivative was used in a similar manner to achieve mechanically induced fluorescence turn-on. On account of an excited state intramolecular proton transfer (ESIPT), the hydroxy-benzoxazole is photoluminescent, but esterification affords to a 'caged' molecule, in which the ESIPT is suppressed. Thus, a benzoxazole ester was used to initiate the polymerization of methyl acrylate through an atom transfer radical polymerization, affording poly(methyl acrylate) comprising a chain-centered caged luminophore. Ultrasonication experiments were used to induce mechanochemical chain cleavage of the benzoxazole ester, which in turn restores the photoluminescence of the patent hydroxy-benzoxazole.