Triazolylidene iron(II) piano-stool complexes: synthesis and catalytic hydrosilylation of carbonyl compounds
1,2,3-Triazolylidenes are a recently developed sub-class of NHC ligands which have tremendous versatility due to the synthetic flexibility of the copper catalysed cycloaddition of alkynes with azides (CuAAC).1,2 These ligands are strong σ-donors, exhibiting stronger donor properties than classic Arduengo-type imidazole-2-ylidenes.2 This property, coupled with the electronic flexibility of the mesoionic ligands makes them a powerful class of ligands for a large variety of catalytic transformations.
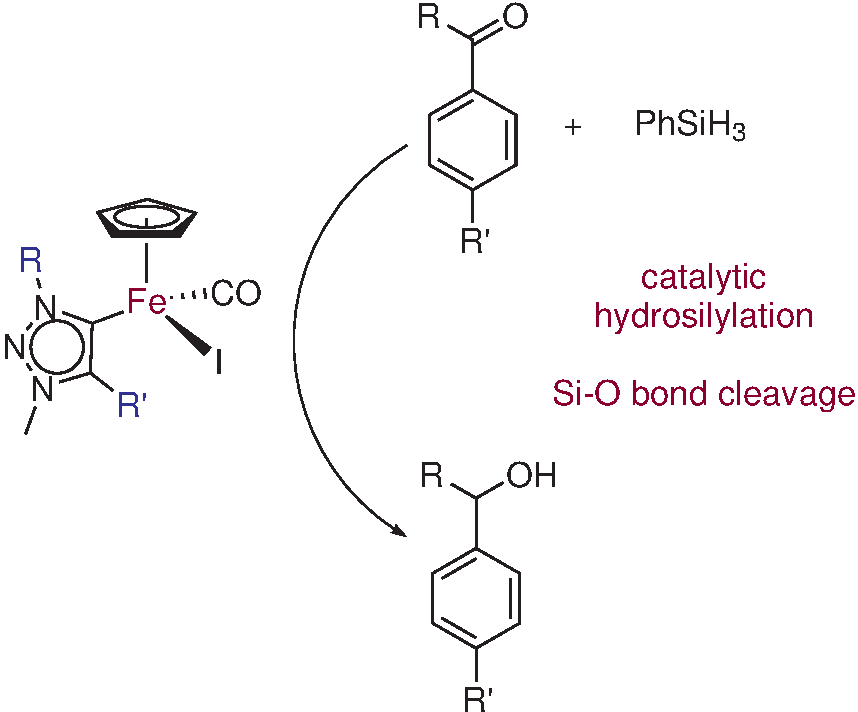
Iron is inexpensive, earth-abundant, non-toxic, biologically relevant, and environmentally benign. Thus, iron catalysts offer an attractive alternative to the systems based on rare and precious platinum group and coinage metals which dominate current literature.3 Herein we present a new class of triazolylidene iron(II) piano stool complexes and their activity in catalytic hydrosilylation reactions.
[1] P. Mathew, A. Neels, M. Albrecht, J. Am. Chem. Soc., 2008, 130, 13534-13535.
[2] K. F. Donnelly, A. Petronilho, M. Albrecht, Chem. Commun., 2013, 49, 1145-1159.
[3] K. Riener; S. Haslinger, A. Raba, M. P. Högerl, M. Cokoja, W. A. Herrmann, F. E. Kühn, Chem. Rev., 2014, 114, 5215-5272.