The History of “BLORANGE”: Co-Sensitized Copper(I) Dye-Sensitized Solar Cells
Renewable energy sources are the answer to save the world from drastic climate changes. The most promising one is photovoltaics. Today, photovoltaic modules are based on silicon but also consist rare elements. A very good alternative are dye-sensitized solar cells, which had its breakthrough in 1991 [1]. The most efficient cells contain ruthenium which is also rare on earth.
Our group focuses its research on the application of copper(I) complexes in dye-sensitized solar cells [2]. A dye-sensitized solar cells consists of different components, which can be optimized to yield higher efficiencies.
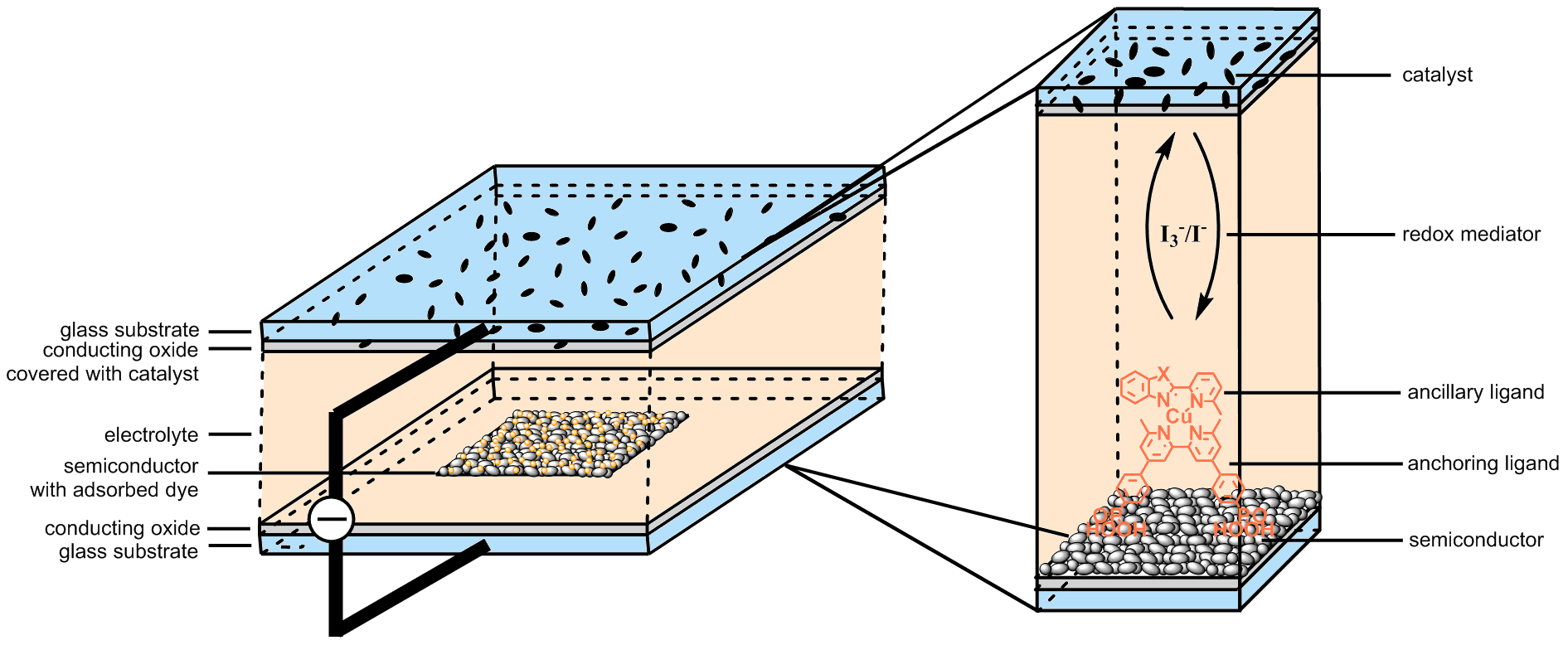
Figure 1: Structure of a dye-sensitized solar cell with zoom-in example for investigated copper(I) dye-sensitized solar cells.
The most important part of a dye-sensitized solar cell is the dye. It consists of two parts – the anchoring and the ancillary ligand [3]. In case of a copper(I) dye-sensitized solar cell the absorption maximum is at about 480 nm and the photon-to-electricity conversion efficiency between 600 and 700 nm is rather low. To reach a panchromatic absorption, co-sensitization with another dye is sufficient. The relatively high efficiencies show the huge potential of co-sensitized copper(I) dye-sensitized solar cells.
[1] B. O’Regan and M. Grätzel, Nature, 1991, 353, 737.
[2] C. E. Housecroft and E. C. Constable, Chem. Soc. Rev., 2015, 44, 8386.
[3] F. J. Malzner, C. E. Housecroft, E. C. Constable and M. Willgert, J. Mater. Chem. A, 2017, 5, 4671.