Quinones as Reversible Electron Relays in Artificial Photosynthesis
The ultimate source of electrons for photocatalytic hydrogen production is water. The four-electron oxidation of two H2O to O2 involves several steps associated with a high kinetic overpotential. So far, this prevented its widespread use as an electron donor in full, molecular water splitting systems. Therefore, the two half-reactions in artificial photosynthesis are commonly studied using sacrificial agents. On the reductive side, irreversible or semi-reversible electron donors such as tertiary amines or ascorbic acid were applied.[1,2] This practice was established to study the half-reaction individually, without interference of the oxidative side. However, this concept has little practical significance.
In contrast, nature uses a completely reversible electron relay in the form of the hydroquinone/quinone system to shuttle electrons between PSII and PSI.[3] Compared to the oxidized form of tertiary amines and ascorbate, oxidized hydroquinones are excellent electron acceptors, thus efficiently inhibiting any productive electron transfer reactions in the reductive half-reaction to H2. A photocatalytic proton reduction system featuring a reversible hydroquinone/quinone shuttle system that overcomes this limitation will be presented along with a kinetic analysis of the system elucidating the faith of the quinone species as well as the bottle neck of the system.[4]
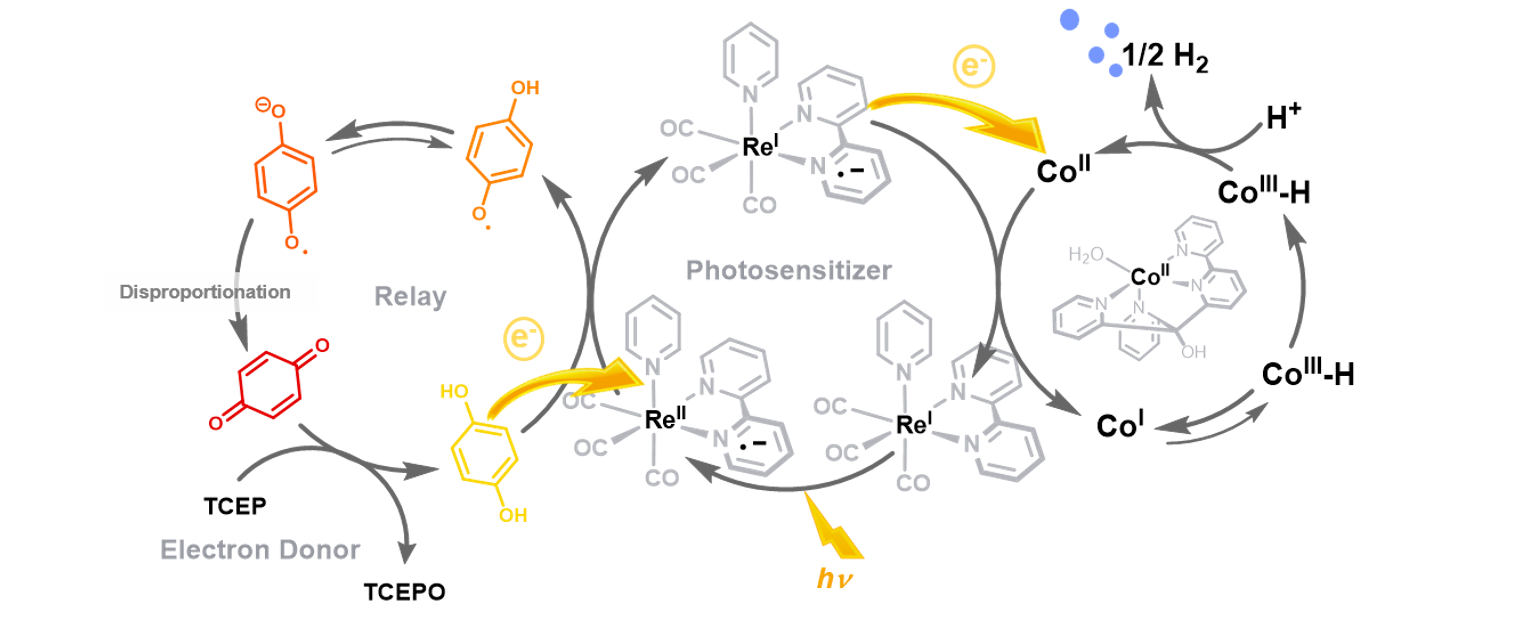
Fig. 1: Reductive half reaction of artificial photosynthesis with TCEP as sacrificial electron donor, a quinone-based relay, a Ru-photosensitizer and a Co-based water reduction catalyst (WRC).
[1] B. Probst, M. Guttentag, A. Rodenberg, P. Hamm, R. Alberto, Inorg. Chem., 2011, 50, 3404–3412.
[2] M. Guttentag, A. Rodenberg, B. Probst, P. Hamm, R. Alberto et al, Eur. J. Inorg. Chem., 2012, 2012, 59–64.
[3] S. Berardi, M. Guttentag, C. Richmond, T. Stoll, A. Llobet et al, Chem. Soc. Rev., 2014, 43, 7501–7519.
4] A. Rodenberg, M. Orazietti, M. Mosberger, R. Alberto, P. Hamm et al, ChemPhysChem, 2016, 17, 1321–1328.