Homochiral recognition and excellent atroposelectivity in the oxidative coupling of cationic [4]helicenes
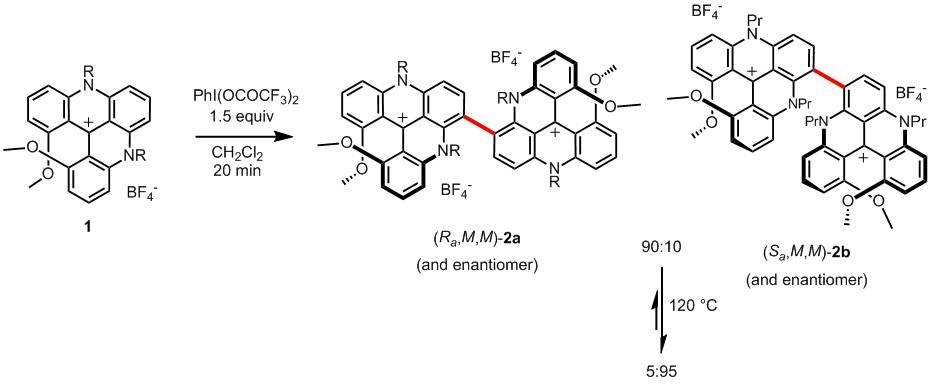
Homochiral recognition by non-covalent interactions is a rather common phenomenon in helical structures.[1] However, this asymmetric trend has been hardly exploited for the formation of covalent bonds between helicenes.[2] Herein, we report the oxidative coupling of the cationic [4]helicenes 1, which proceeds with a quasi exclusive homochiral recognition (96% selectivity) to yield chiral (racemic) dimers of type 2. The lack of meso isomers is established by HRMS cross-over experiments. Moreover, this reaction is highly atroposelective forming preferentially (Ra,M,M)-2a over (Sa,M,M)-2b (and enantiomers) (ratio 90:10). The interconversion barrier from 2a to 2b is 26.4 kcal/mol at 60 °C in dmso-d6. Strong chiroptical properties are observed, for both 2a and 2b, in the red visible region.
[1] Ryo Amemiya, Masahiko Yamaguchi, Organic and Biomolecular Chemistry, 2008, 6, 26-35. Emmanuel Anger, Hiroki Iida, Tomoko Yamaguchi, Koutarou Hayashi, Daisuke Kumano, Jeanne Crassous, Nicolas Vanthuyne, Christian Roussel, Eiji Yashima, Polymer Chemistry 2014, 5, 4909-4914.
[2] Daisuke Nakano, Rie Hirano, Masahiko Yamaguchi, Chizuko Kabuto, Tetrahedron Letters, 2003, 44, 3683-3686.