Kinetics of Rh(II)-Catalyzed α-Diazo-β-Ketoester Decomposition for Polyether Macrocycle Synthesis and Straightforward Access to Ditopic Cryptands
Previously, our group reported the synthesis of polyether macrocycles 1 by [3+6+3+6] condensations of α-diazo-β-ketoesters 2 and 1,4-dioxane under dirhodium catalysis at 1 M concentration.[1] By reactions of 1 with aromatic amines under basic conditions, chiral crown ethers 3 can then be obtained in a single step by tandem amidation / olefin transposition.[2] These compounds 3 are effective pH-insensitive nanosensors and ratiometric luminescent switches.[3] For these and other applications, preparation of 1 in large quantities was required. Kinetics of decomposition of diazo 2 with various rhodium(II) catalysts and different amounts of dioxane were studied by in situ FT-IR monitoring. These mechanistic results showed the superior activity of Hashimoto-Ikegami-like catalyst 4. Reaction conditions were optimized leading to a decrease of catalyst loading (down to 0.001 mol%) and a scale-up of the reaction up to 20 grams of 1 in a single batch.[4]
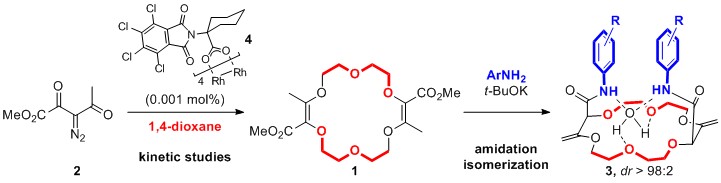
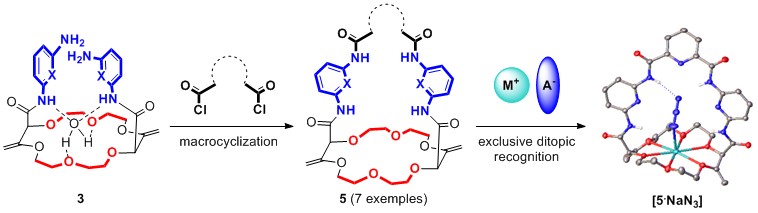
Herein, we present in addition a new family of cryptands 5 readily synthesized in two steps from compounds 1. Hosts 5 display a ditopic character towards sodium salts of linear anions in particular as demonstrated by 1H NMR spectroscopic and solid state structural analysis.
[1] Walid Zeghida, Céline Besnard, Jérôme Lacour, Angewandte Chemie International Edition 2010, 49, 7253-7256.
[2] Mahesh Vishe, Radim Hrdina, Amalia I. Poblador-Bahamonde, Céline Besnard, Laure Guénée, Thomas Bürgi, Jérôme Lacour, Chemical Science 2015, 6, 4923-4928.
[3] a) Zdenka Jarolímová, Mahesh Vishe, Jérôme Lacour, Eric Bakker, Chemical Science 2016, 7, 525-533; b) Stephan Sinn, Frank Biedermann, Mahesh Vishe, Alessandro Aliprandi, Céline Besnard, Jérôme Lacour, Luisa De Cola, ChemPhysChem 2016, 17, 1829-1834.
[4] Daniele Poggiali, Alexandre Homberg, Thimothée Lathion, Claude Piguet, J. Lacour, ACS Catalysis 2016, 6, 4877-4881.