Versatile Remote Functionalizations via C-H/C-C Bond Cleavage Under Mild Conditions
Exploiting inert bonds at non-reactive positions of organic molecules represents both an attractive and equally challenging endeavor. Unique opportunities to streamline the access to structural diversity and complexity can arise from the remote functionalization of C-H and C-C bonds present in distant positions to a functional group, which has fostered major efforts in this area.[1] Here, we describe a radical-mediated, directing-group-free regioselective 1,5-hydrogen transfer of unactivated Csp3-H bonds followed by a Csp2-H functionalization to produce, with exquisite stereoselectivity, a variety of elaborated fused ketones, which demonstrates that aliphatic acids can be strategically harnessed as 1,2-diradical synthons and that secondary aliphatic C-H bonds can be engaged in stereoselective C-C bond forming reactions.[2] We also present a redox-neutral, light-mediated functionalization of unactivated Csp3-H bonds via iminyl radicals, to access divergent scaffolds via Csp3-N or Csp3-Csp2 bond formation upon judicious choice of the reaction conditions.[3] On the other hand, we demonstrate the cleavage of unstrained C-C bonds under mild, visible-light mediated, redox neutral conditions. In situ generated C-centered radicals can be harvested in the presence of Michael acceptors, thiols and alkyl halides to realize diverse γ-functionalizations by forming new Csp3-Csp3, -H and –Br bonds, respectively.[4]
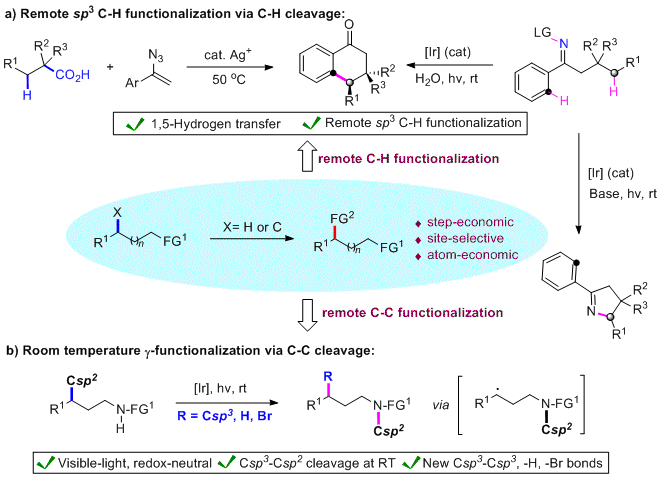
[1] a) R. Breslow, Acc. Chem. Res. 1980, 13, 170. b) H. Schwarz, Acc. Chem. Res. 1989, 22, 282. c) T. Newhouse, P. S. Baran, Angew. Chem. Int. Ed. 2011, 50, 3362. d) A. Vasseur, J. Bruffaerts, I. Marek, Nat. Chem. 2016, 8, 209.
[2] W. Shu, A. Lorente, E. Gómez-Bengoa, C. Nevado, Nat. Commun. 2017, 8, 13832.
[3] W. Shu, C. Nevado, Angew. Chem. Int. Ed. 2017, 56, 1881.
[4] W. Shu, A. Genoux, Z. Li, C. Nevado, Angew. Chem. Int. Ed. 2017, under revision.