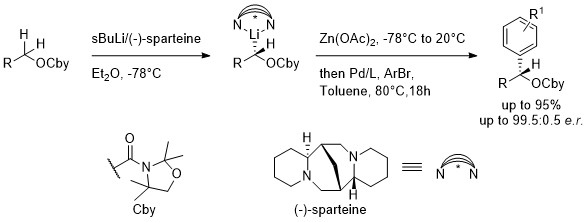
Enantioselective α-Arylation of O-Carbamates via Sparteine-Mediated Lithiation and Negishi Cross-coupling
The enantioselective α-arylation of protected aliphatic alcohols is described. Hoppe’s technology allows to perform the enantioselective α-lithiation in presence of sparteine. [1] After Li-Zn transmetalation and Negishi cross-coupling, highly enantioenriched benzylic alcohols are accessed. The method is compatible with a wide range of (hetero)aryl bromides and aliphatic alcohols.
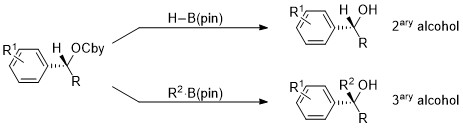
Application of Aggarwal’s lithiation-borylation sequence [2] provides a short and divergent access to a variety of enantioenriched secondary and tertiary benzylic alcohols. [3]
[1] a) Beak, P.; Lee, K. W. J. Org. Chem. 1993, 58, 1109-1117. b) Hoppe, D.; Hense, T. Angew. Chem. Int. Ed. 1997, 36, 2282-2316.
[2] Stymiest, J. L.; Bagutski, V.; French, R. M.; Aggarwal, V. K. Nature 2008, 456, 778-782.
[3] Royal, T.; Baumgartner, Y.; Baudoin, O. Org. Lett. 2017, 19, 166-169.